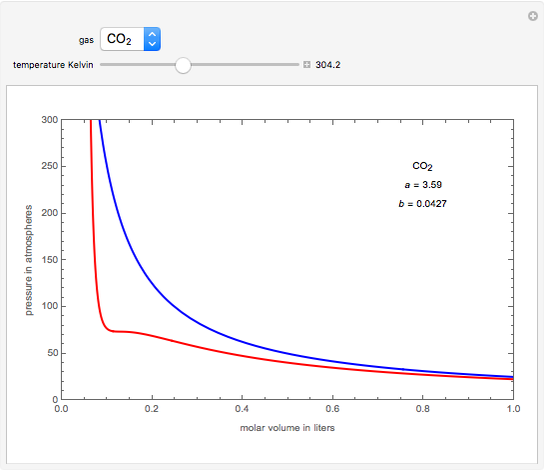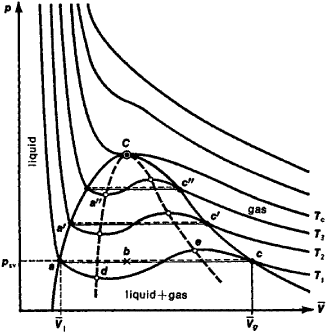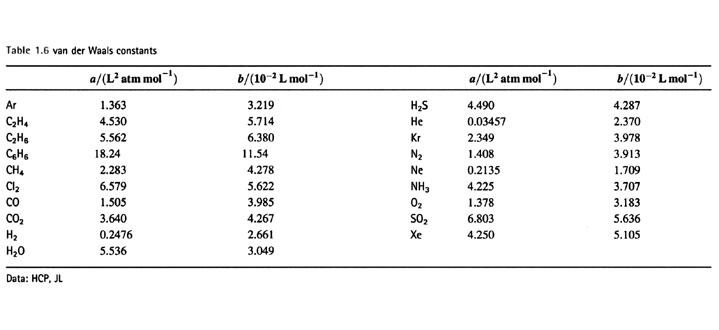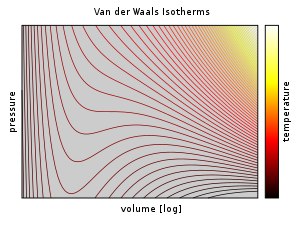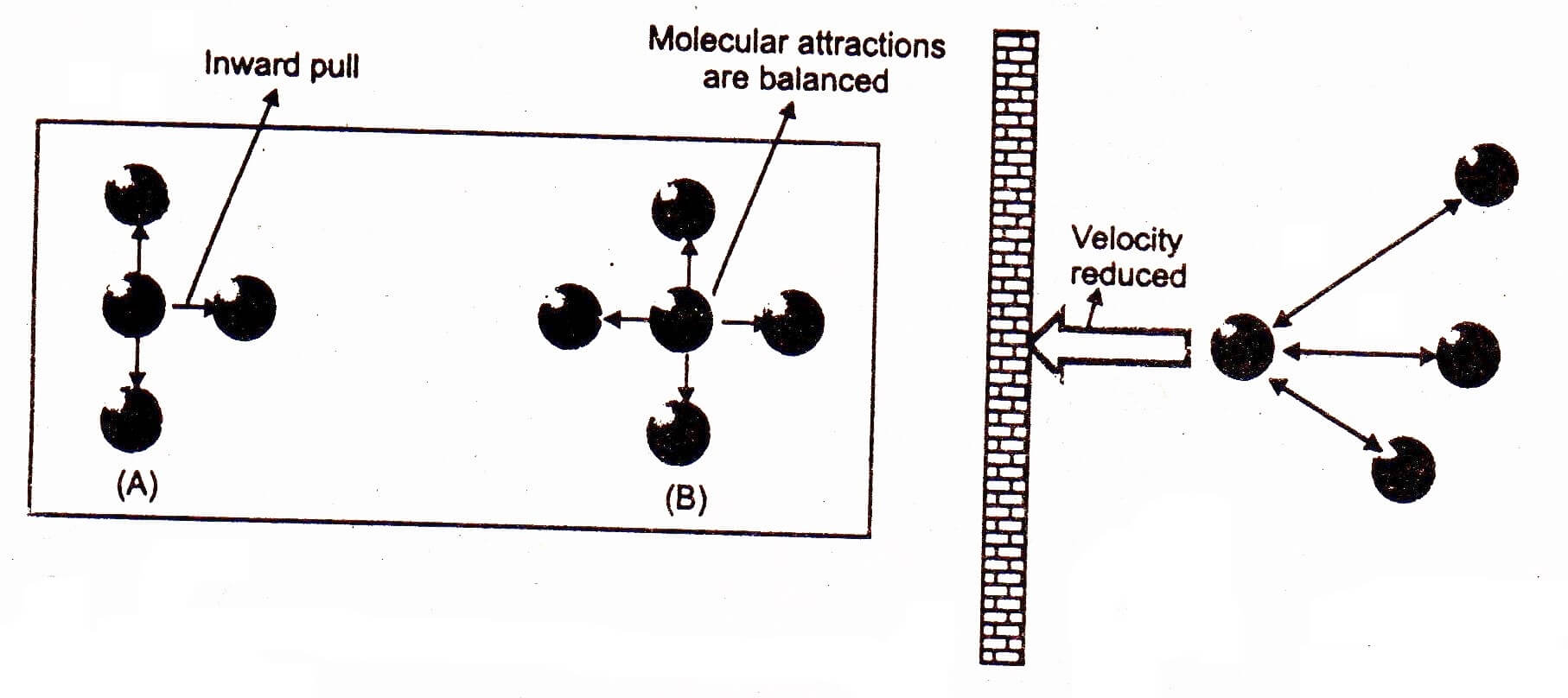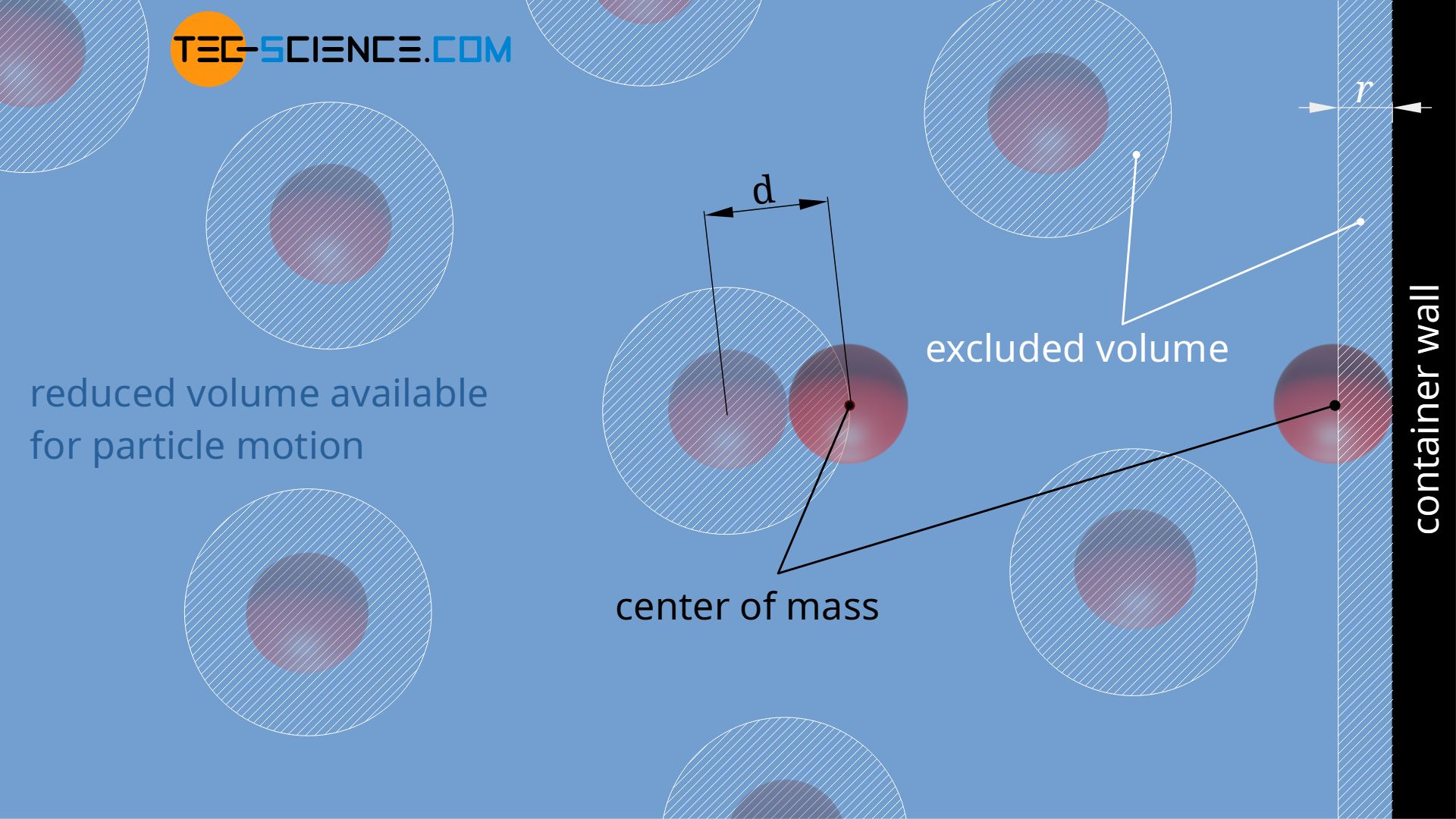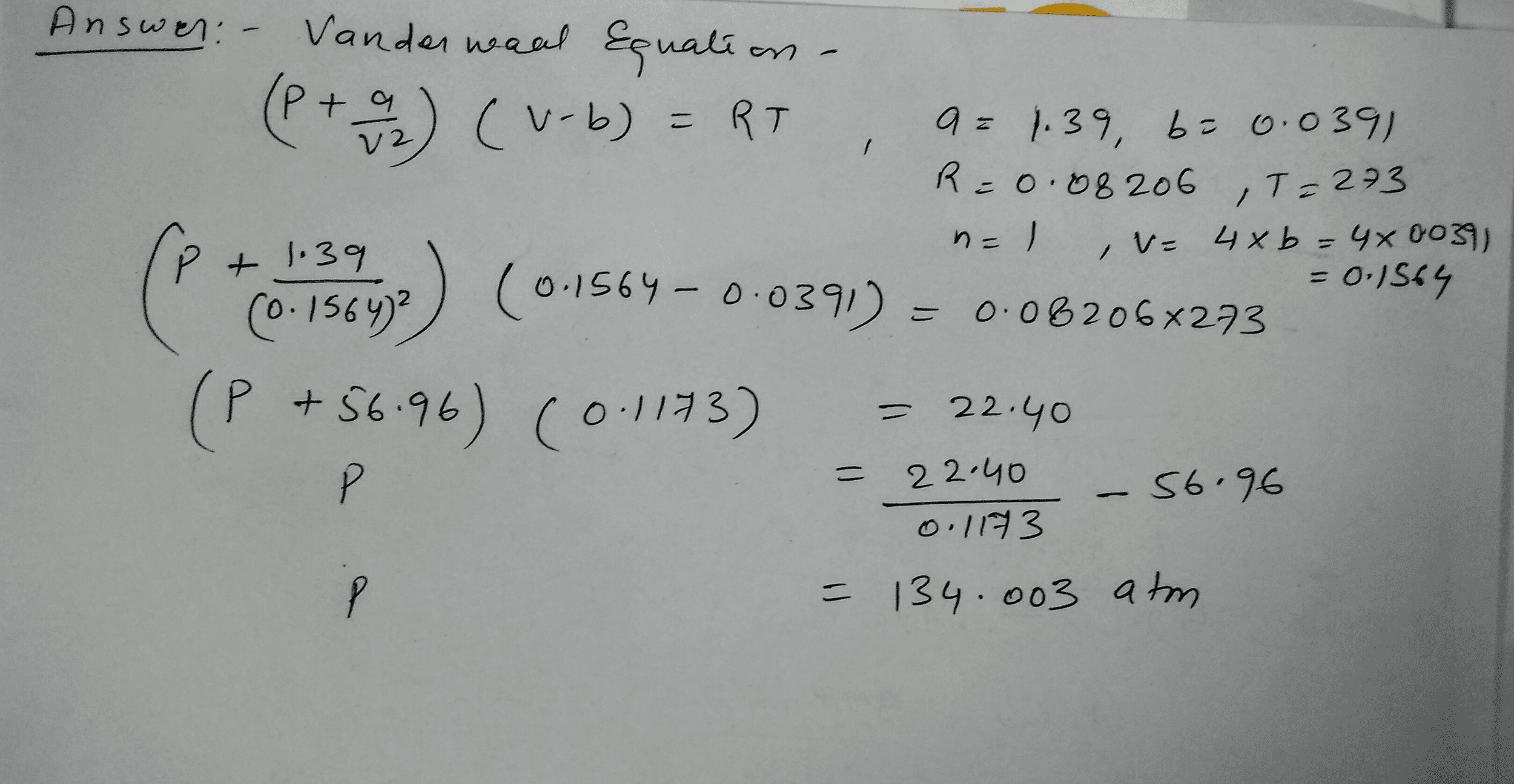
OneClass: Use the van der waals equation of state to calculate the pressure of N2 at which its molar ...
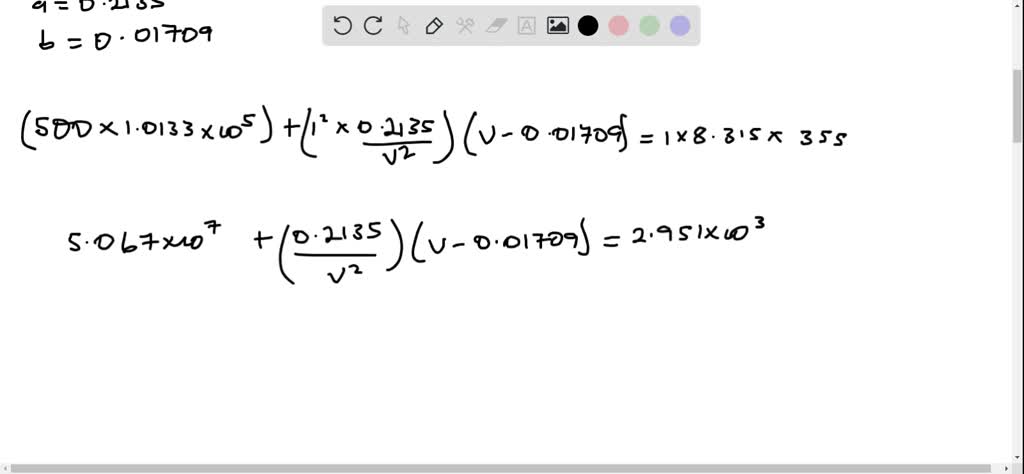
SOLVED: Use the van der Waals equation and the ideal gas equation to calculate the volume of 1.000 mol of neon at a pressure of 500.0 atm and a temperature of 355.0