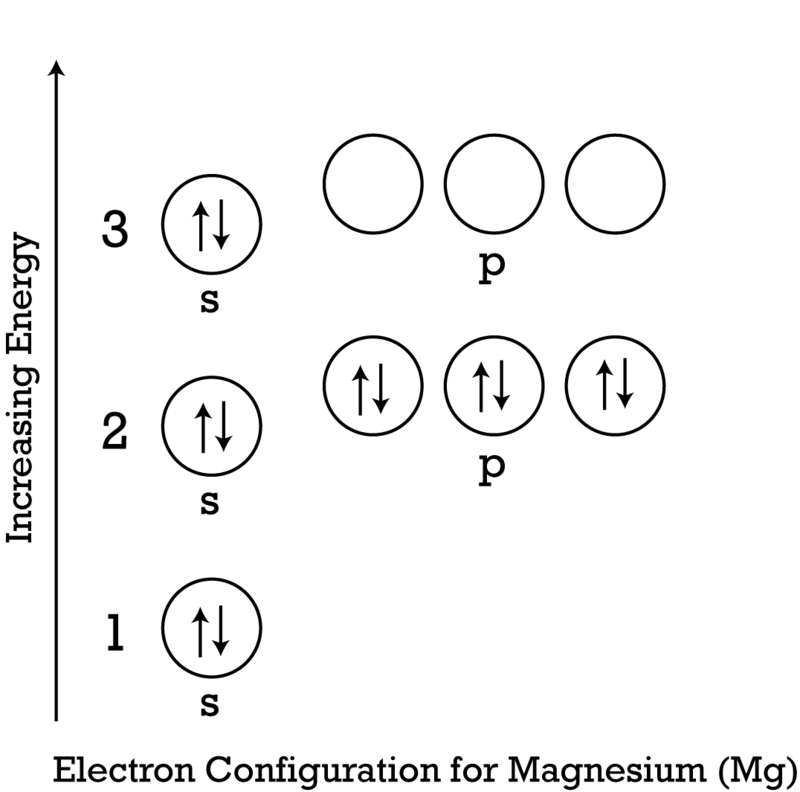
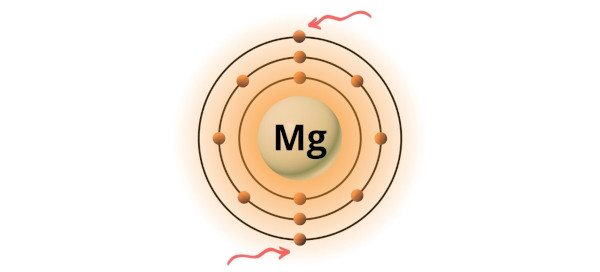
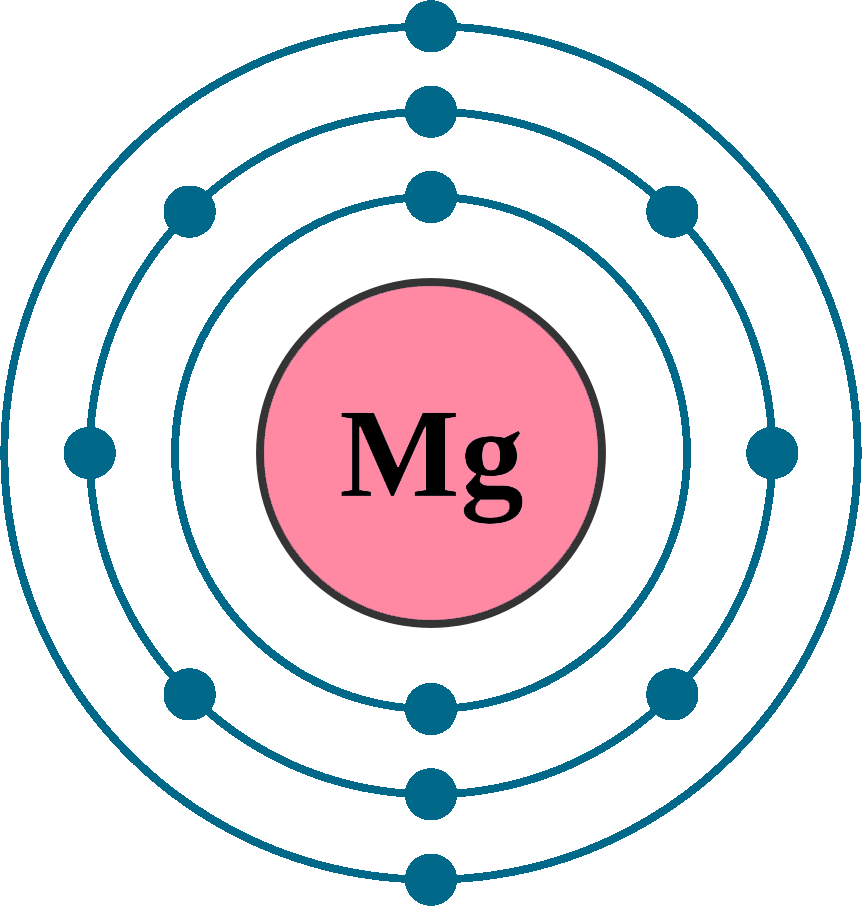
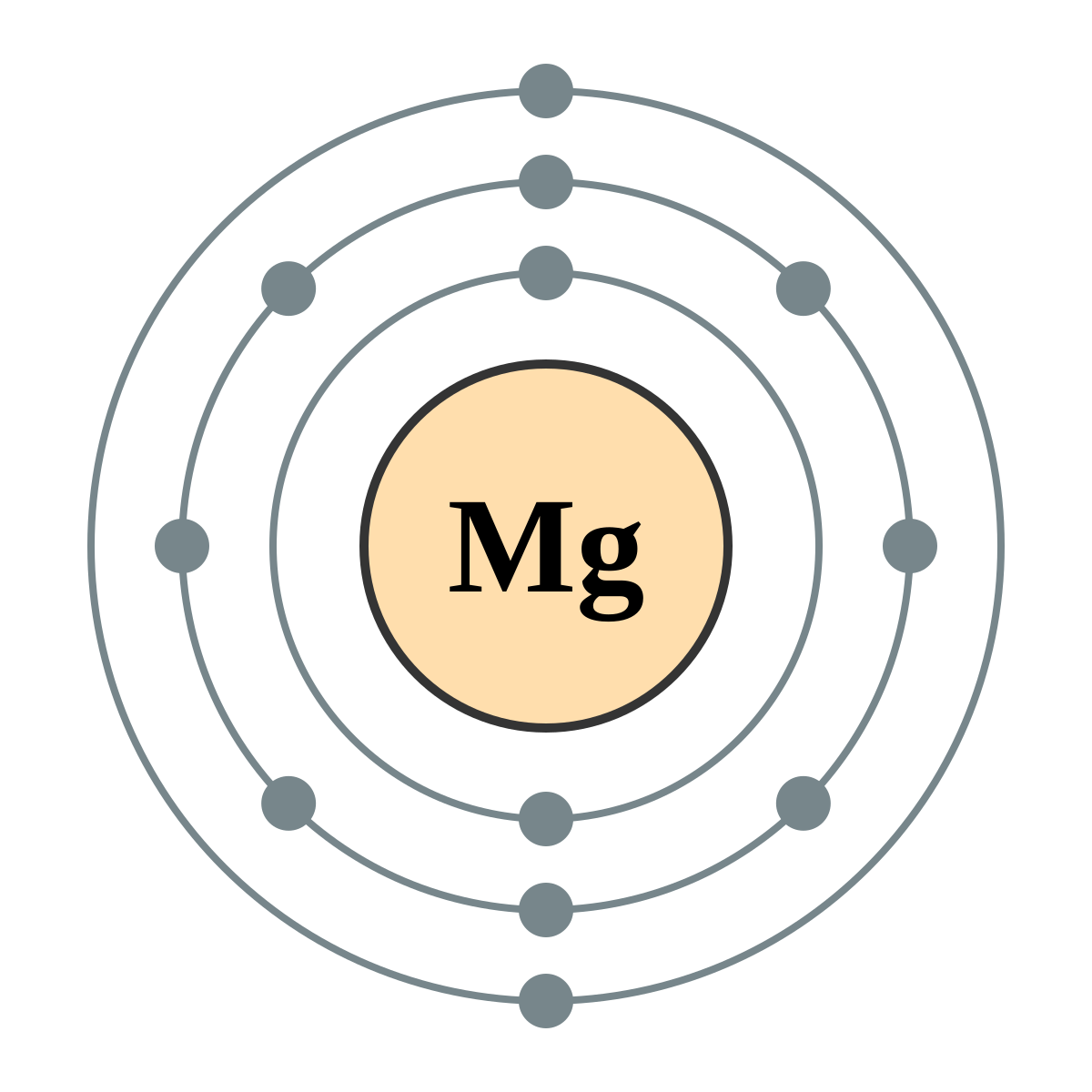
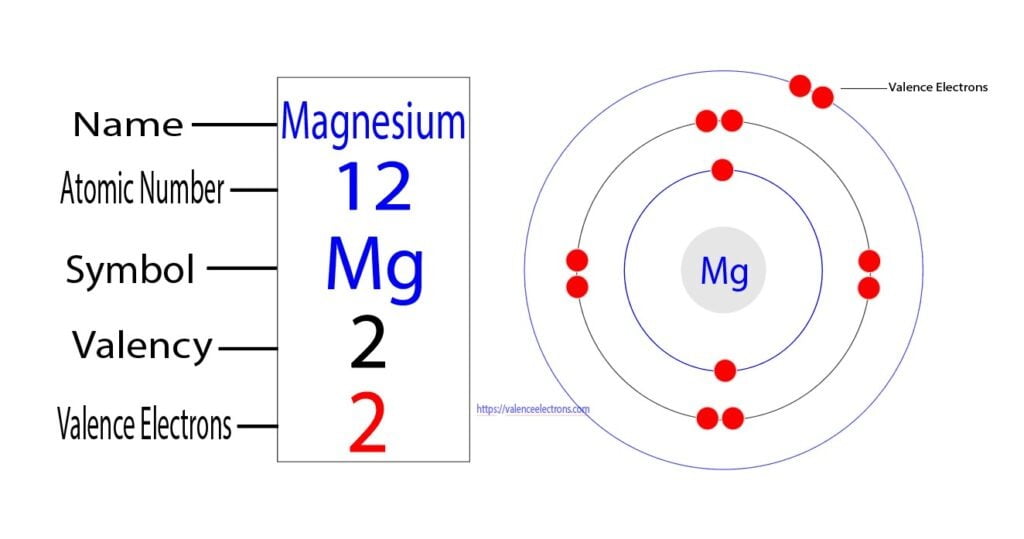
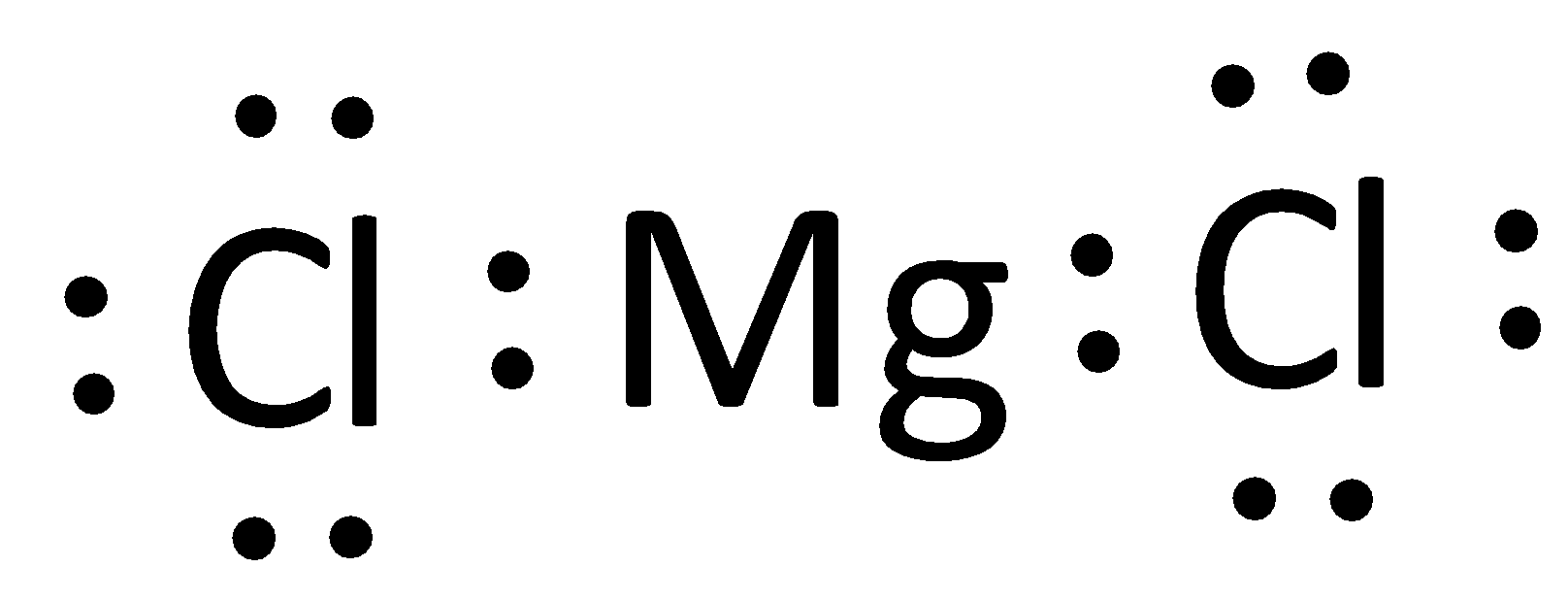Represent the transfer of electrons from magnesium to oxygen atoms to assume noble-gas configurations. Use Lewis electron-dot symbols. | Homework.Study.com

How Many Valence Electrons Does Magnesium (Mg) Have?||Number of Valence Electrons in Magnesium (Mg)? - YouTube

Magnesium - Electron Affinity - Electronegativity - Ionization Energy of Magnesium | nuclear-power.com
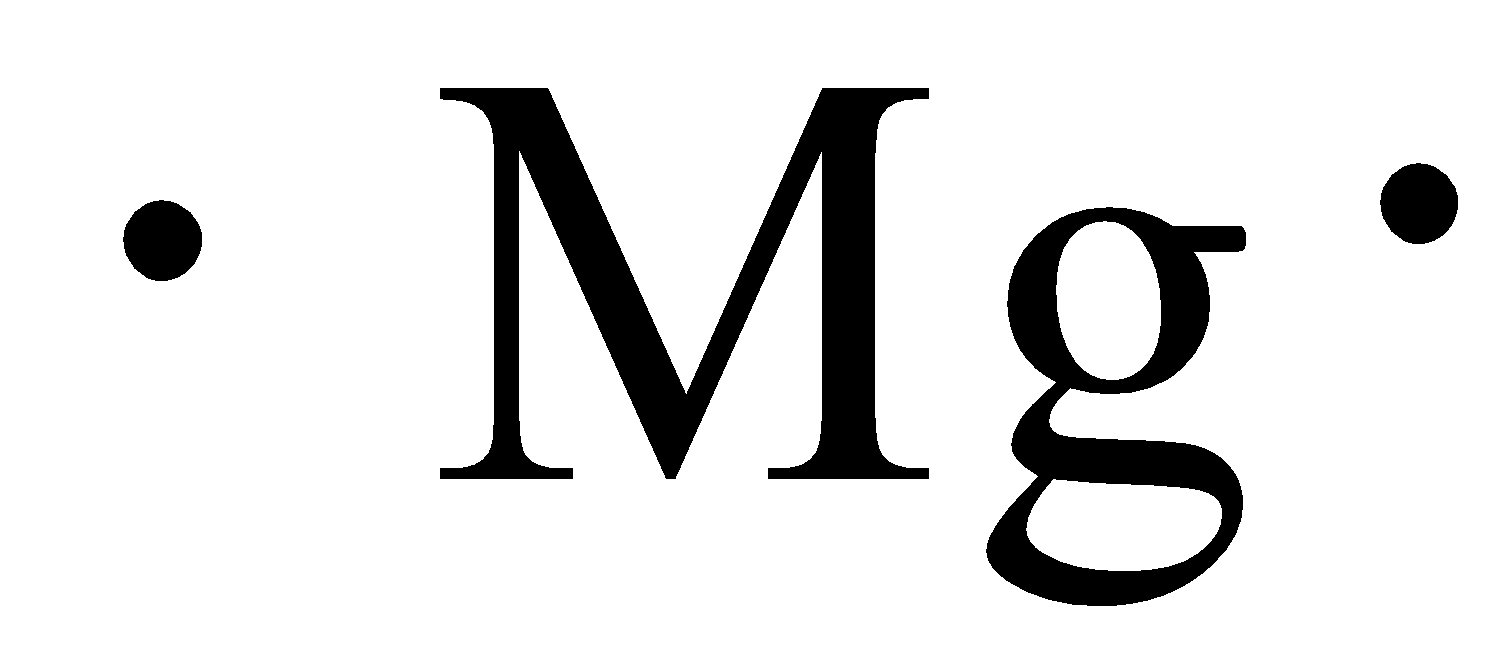
Write the electron dot structure for magnesium and chlorine. Show the formation of magnesium chloride by the transfer of electrons. What are the ions present in this compound?