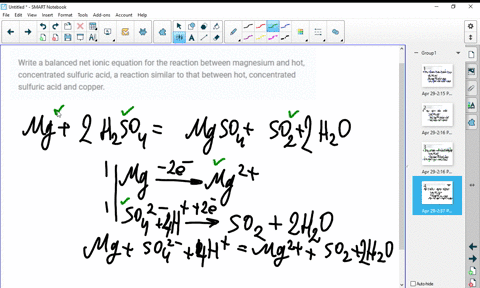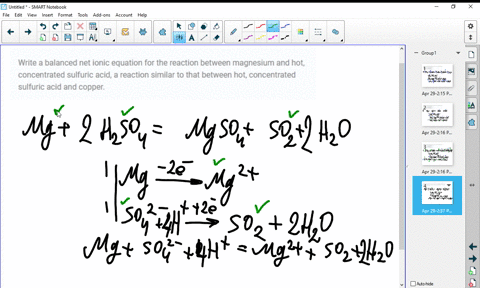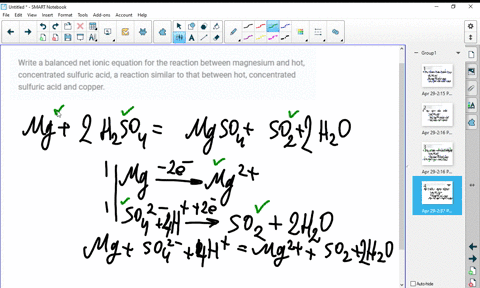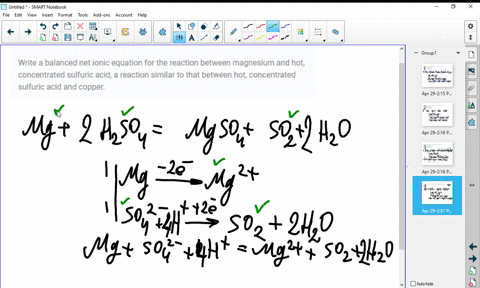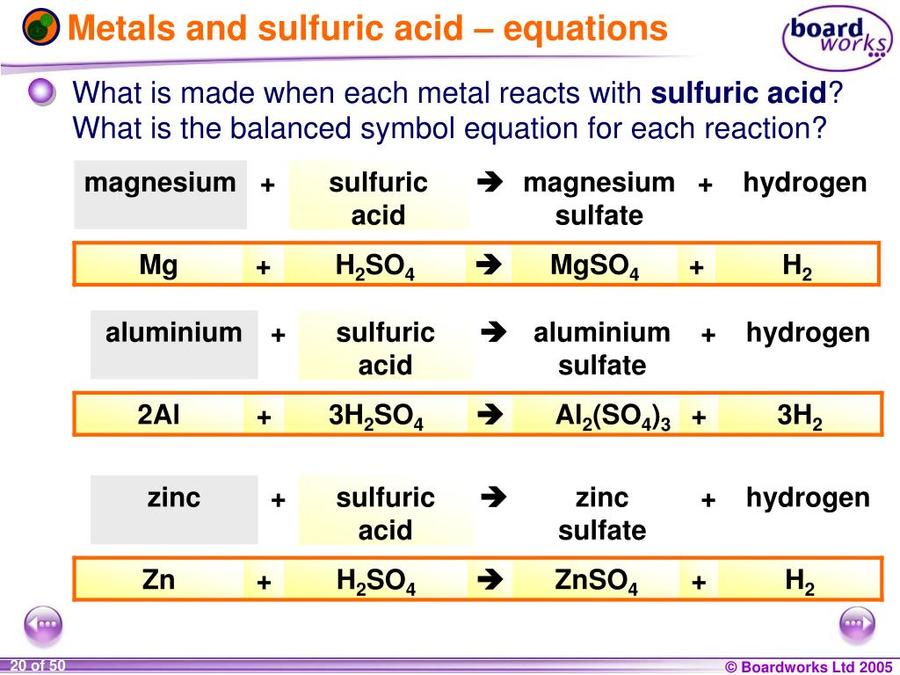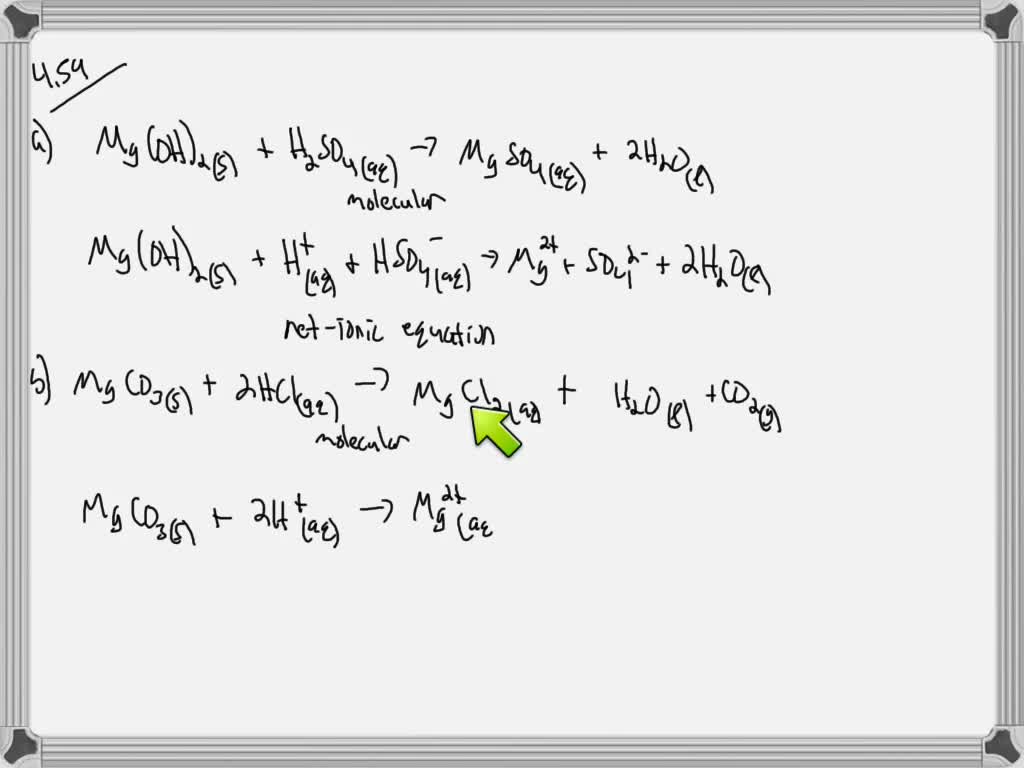
SOLVED:Write a balanced molecular equation and a net ionic equation for the following reactions: a. Solid magnesium hydroxide reacts with a solution of sulfuric acid. b. Solid magnesium carbonate reacts with a

H2SO4+Mg(OH)2=H2O+MgSO4 Balanced Equation|Sulphuric Acid+Magnesium Hydroxide=Water+Magnesium Sulphat - YouTube

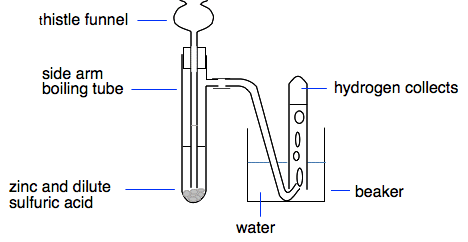
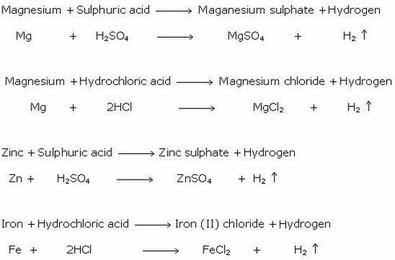
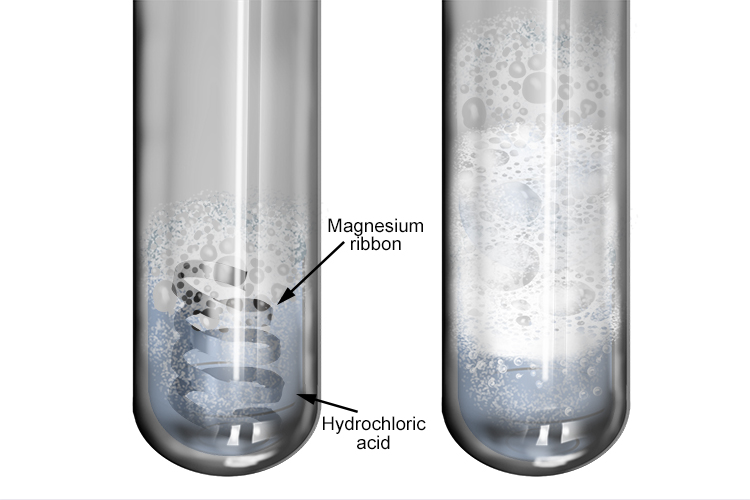
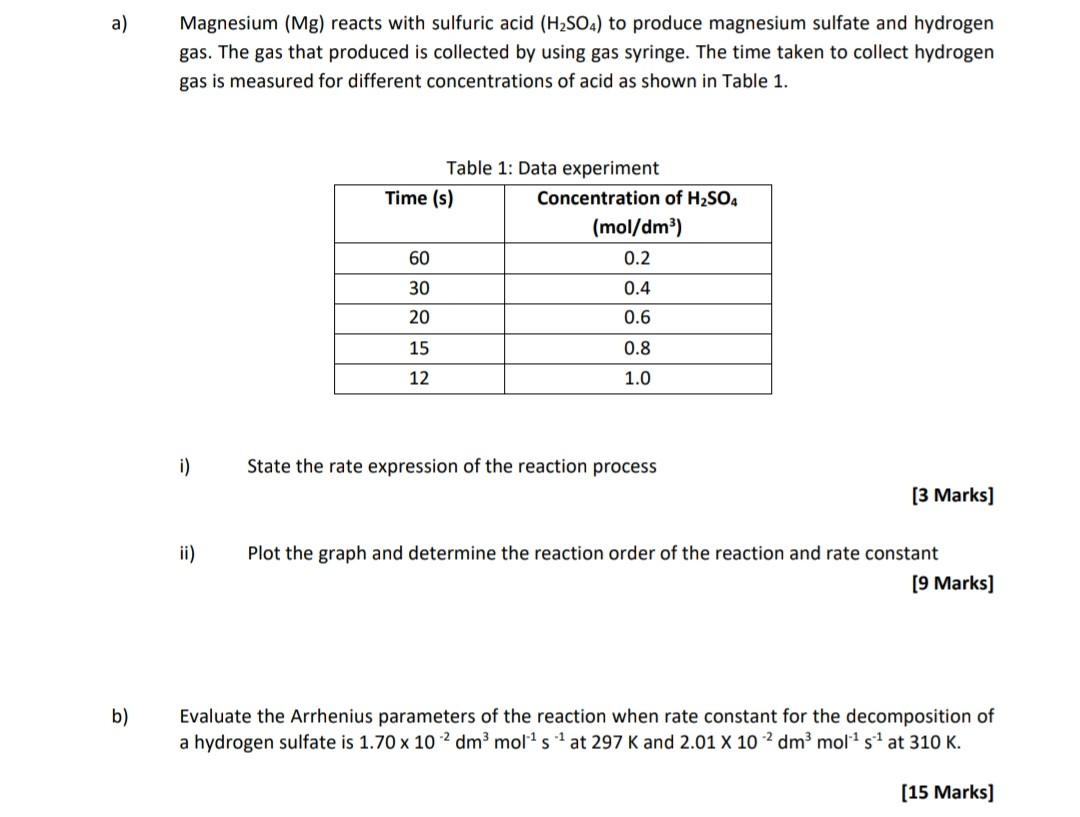
Investigate reactions between dilute hydrochloric and sulphuric acids and metals (eg. Magnesium, Zinc and Iron) Diagram | Quizlet

investigate reactions between dilute hydrochloric and sulfuric acids and metals (e.g. magnesium, zinc and iron) Diagram | Quizlet