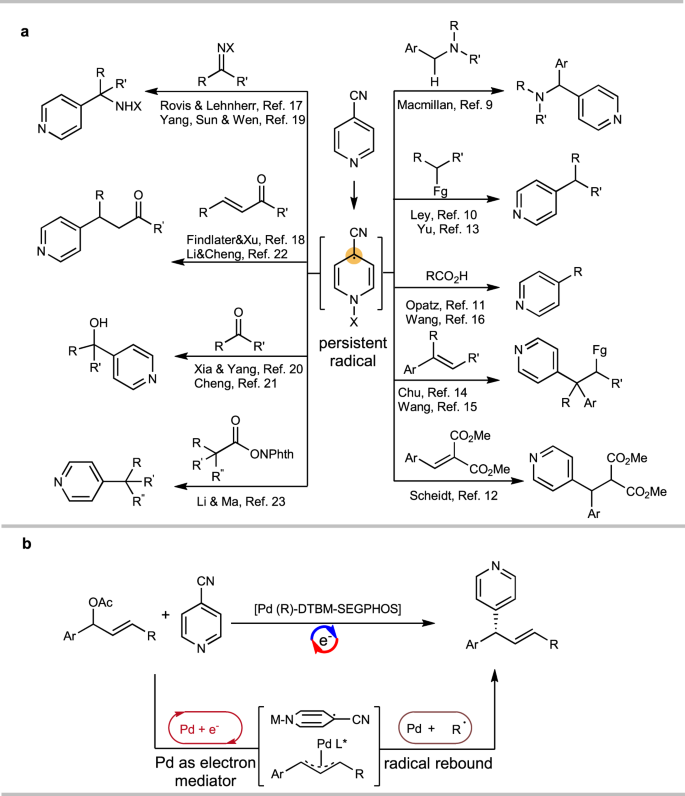
Palladium-catalyzed asymmetric allylic 4-pyridinylation via electroreductive substitution reaction | Nature Communications

Palladium-Catalyzed Deracemization of Allylic Carbonates in Water with Formation of Allylic Alcohols: Hydrogen Carbonate Ion as Nucleophile in the Palladium-Catalyzed Allylic Substitution and Kinetic Resolution | Journal of the American Chemical Society

Palladium-Catalyzed, Carboxylic Acid-Assisted Allylic Substitution of Carbon Nucleophiles with Allyl Alcohols as Allylating Agents in Water | Organic Letters
BASF launches new Tri-Metal Catalyst, enabling partial substitution of palladium with platinum in catalytic converters - Green Car Congress
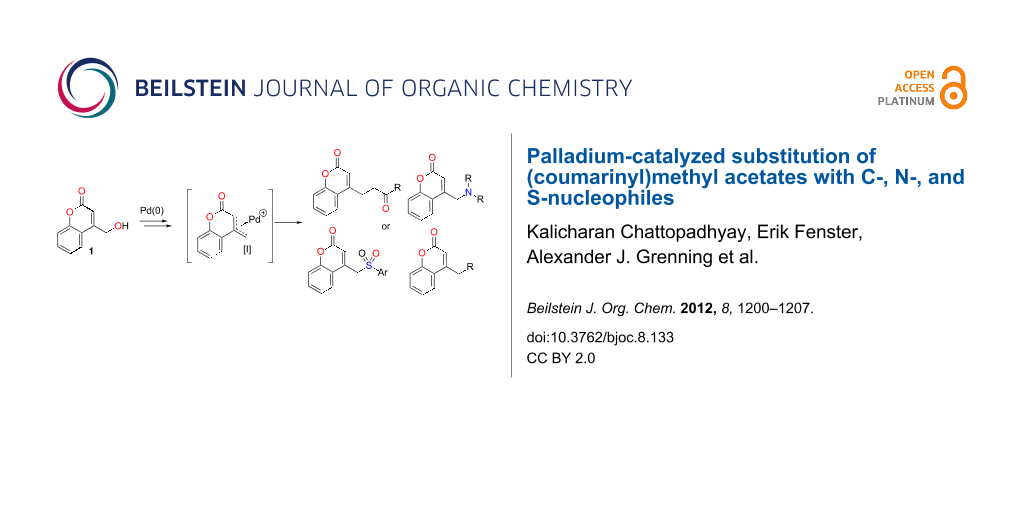
BJOC - Palladium-catalyzed substitution of (coumarinyl)methyl acetates with C-, N-, and S-nucleophiles

Figure 1 from Palladium-catalyzed chemoselective allylic substitution, Suzuki-Miyaura cross-coupling, and allene formation of bifunctional 2-B(pin)-substituted allylic acetate derivatives. | Semantic Scholar

Palladium-Catalyzed Ligand-Controlled Regioselective Nucleophilic Aromatic Substitution of 1-(Chloromethyl)naphthalenes with Arylacetonitriles | The Journal of Organic Chemistry
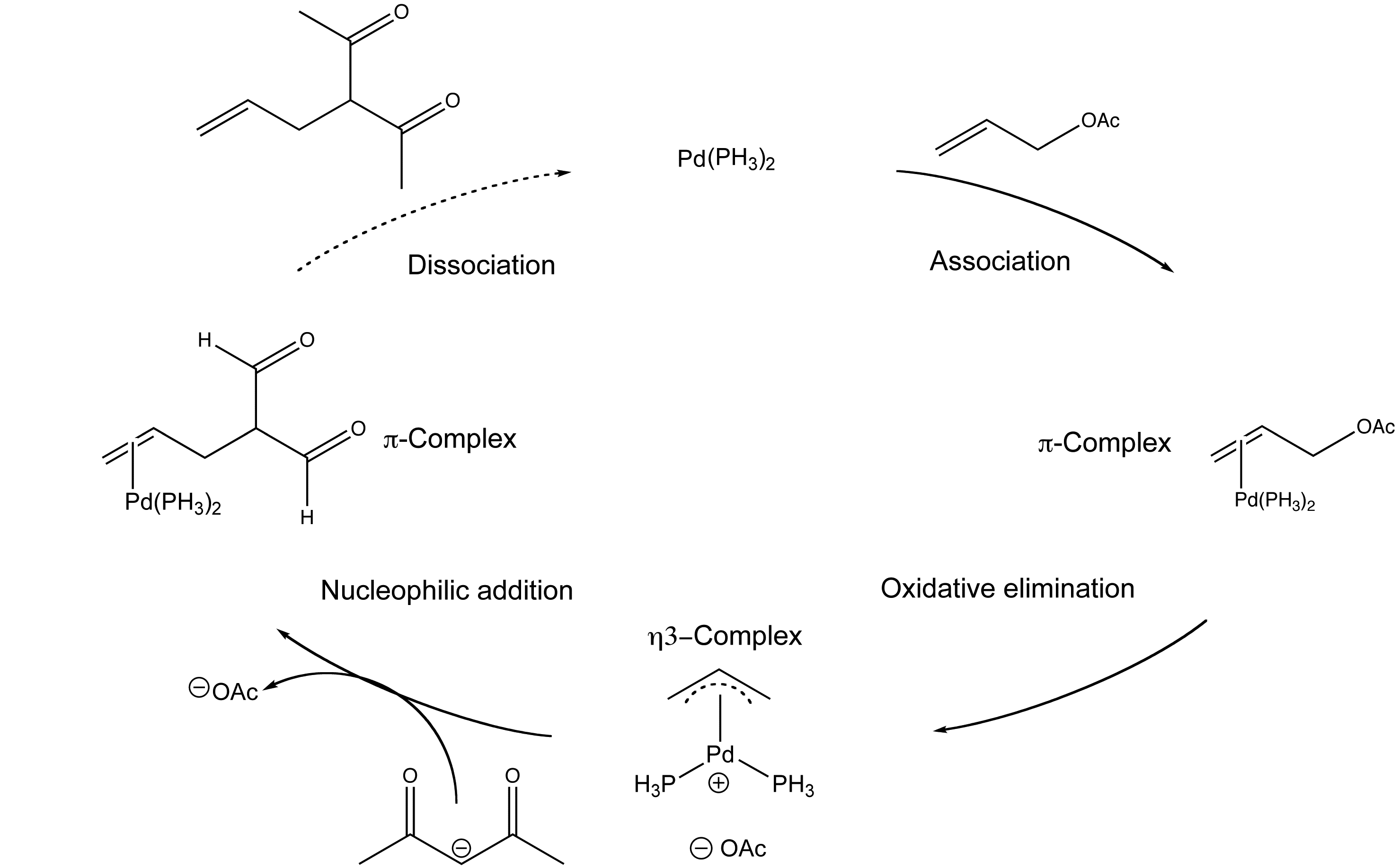
Organopalladium Chemistry - Palladium-catalysed nucleophilic allylic substitution of functionalised compounds

Palladium‐Catalyzed Allylic Substitution for the Synthesis of Pericosines - Chung - 2016 - Asian Journal of Organic Chemistry - Wiley Online Library

Asymmetric palladium-catalyzed nucleophilic substitution of 1-(2-naphthyl)ethyl acetate by dimethyl malonate anion - ScienceDirect

Palladium-Catalyzed, Carboxylic Acid-Assisted Allylic Substitution of Carbon Nucleophiles with Allyl Alcohols as Allylating Agents in Water

Asymmetric allylic substitution by chiral palladium catalysts: Which is more reactive, major π-allyl Pd(II) species or minor π-allyl species? - ScienceDirect
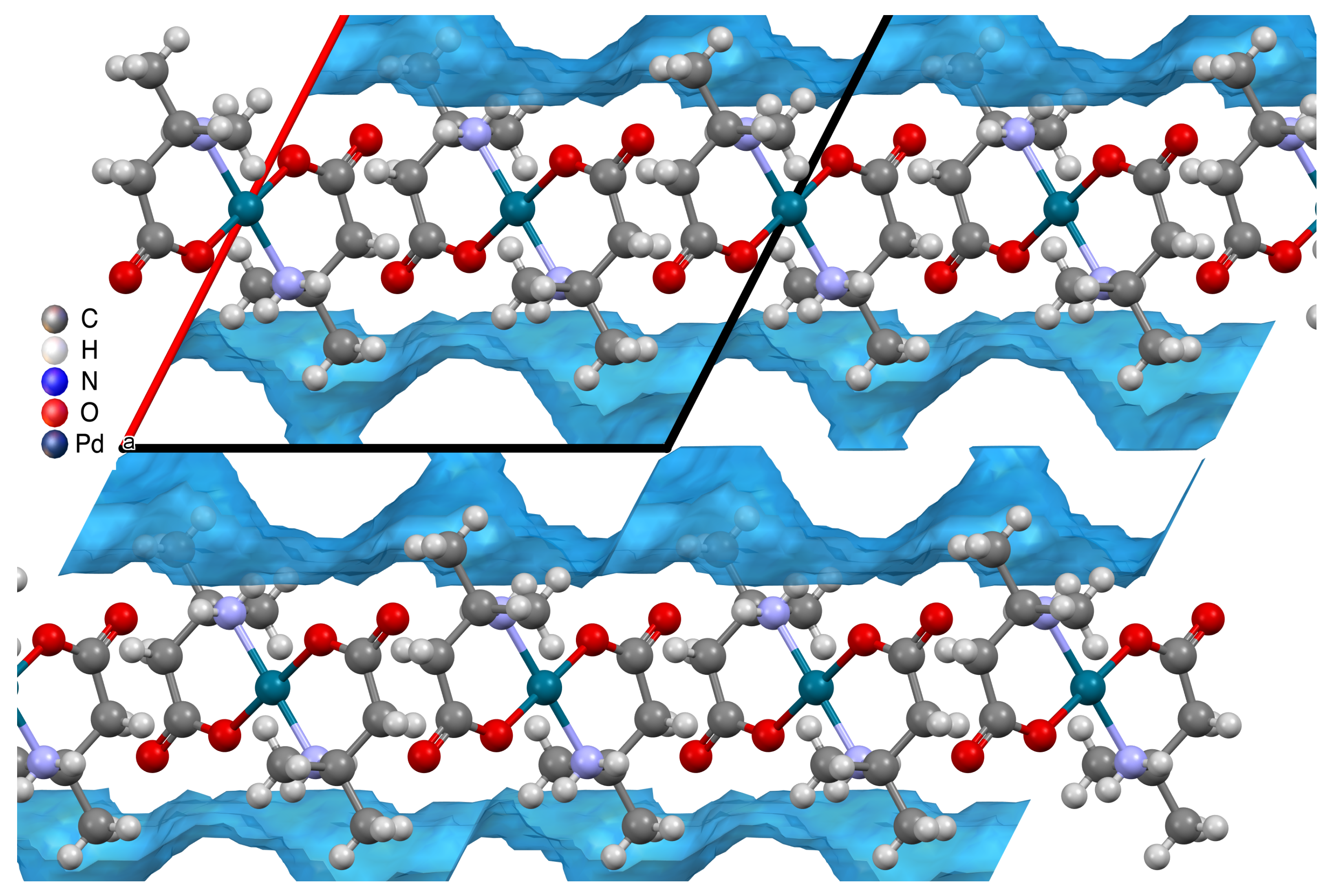
Crystals | Free Full-Text | Self-Assembly Motifs of Water in Crystals of Palladium β-Amino Acid Complexes Influenced by Methyl Substitution on the Amino Acid Backbone

Enantioselective Intramolecular Allylic Substitution via Synergistic Palladium/Chiral Phosphoric Acid Catalysis: Insight into Stereoinduction through Statistical Modeling | Organic Chemistry | ChemRxiv | Cambridge Open Engage
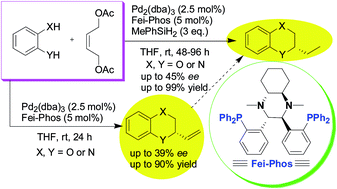
Palladium-catalyzed tandem allylic substitution/cyclization and cascade hydrosilylated reduction: the influence of reaction parameters and hydrosilanes on the stereoselectivity - RSC Advances (RSC Publishing)

Palladium-Catalyzed C−P Bond Formation: Mechanistic Studies on the Ligand Substitution and the Reductive Elimination. An Intramolecular Catalysis by the Acetate Group in PdII Complexes | Organometallics



/cloudfront-us-east-2.images.arcpublishing.com/reuters/2VOUEPFTNJKQZEMHRVLJ4AE4PI.jpg)








