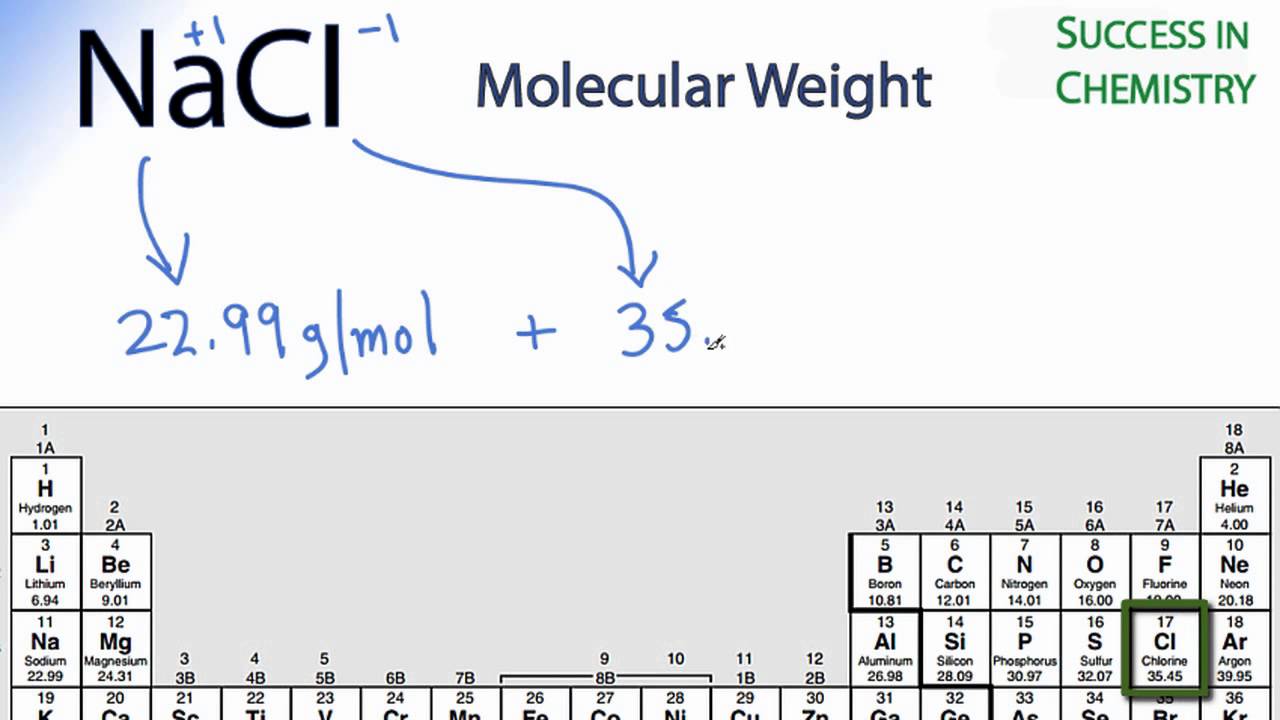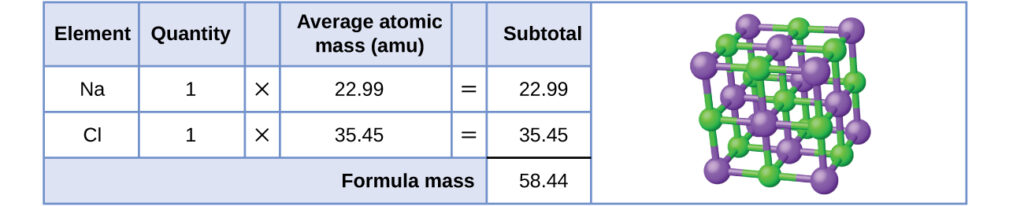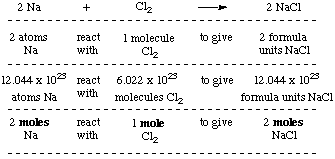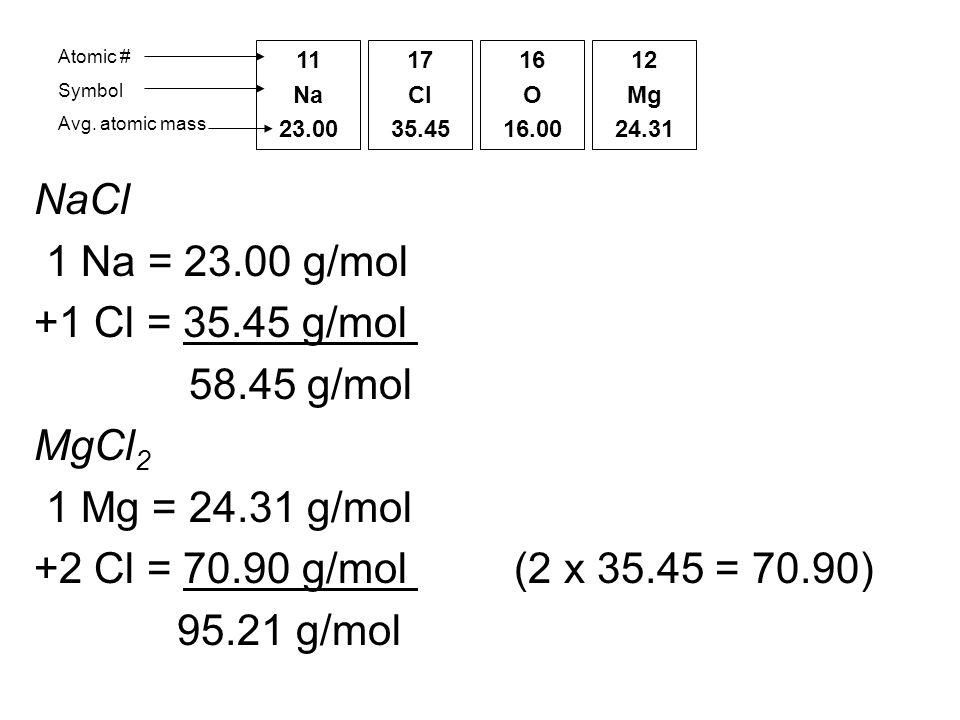
MOLAR MASS Molar mass of a substance = mass in grams of one mole of the substance. A compound's molar mass is NUMERICALLY equal to its formula mass. Formula. - ppt download

Calculate the mass of 1 mole of each one of the following: (a) `NaCl` , (b) `CaCO_(3)` , (c ) `FeSO - YouTube

Table I-B Molar Mass of Some Common CompoundsAtomic Mass of each element in a given compound (g) - Brainly.ph

What you should do:1. For Table 5-B, compute the molar mass of the givencompound. See example number 1 - Brainly.ph



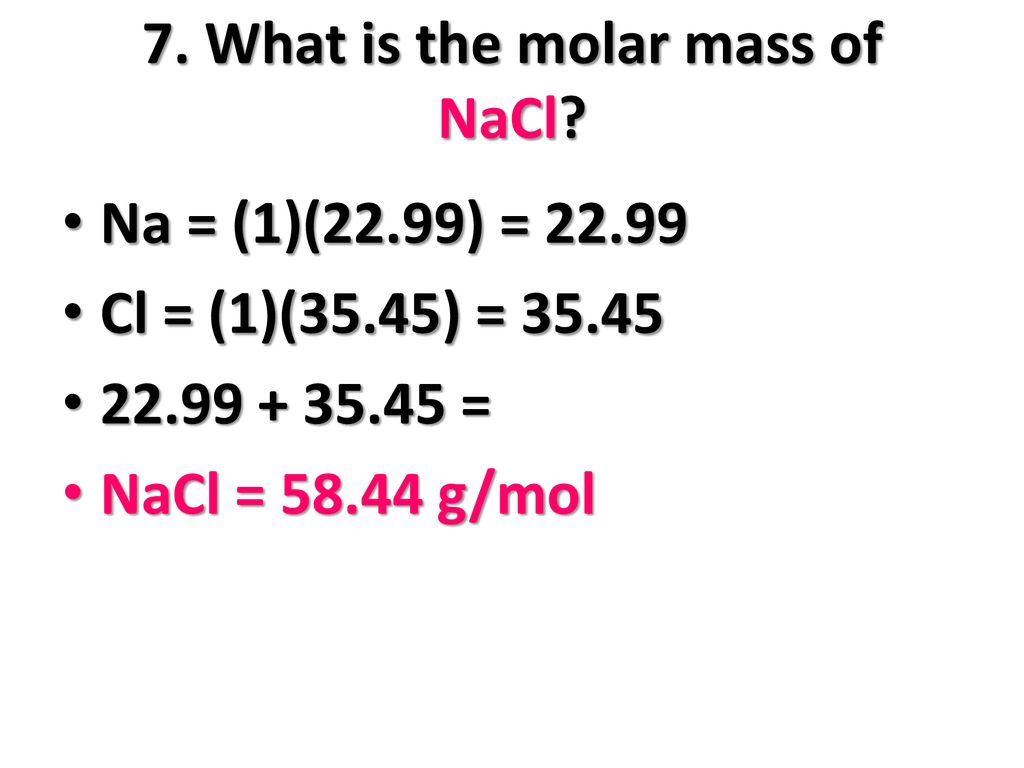


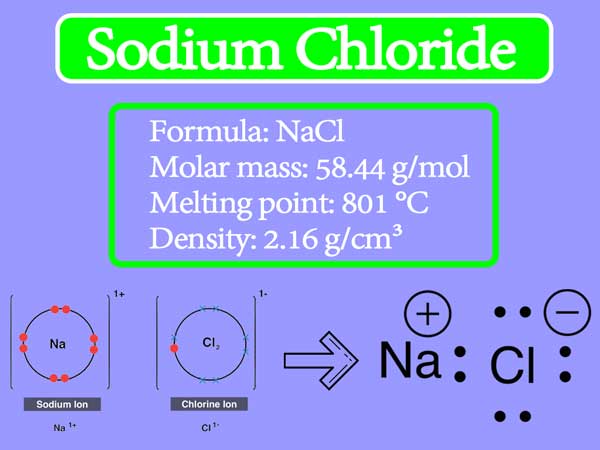






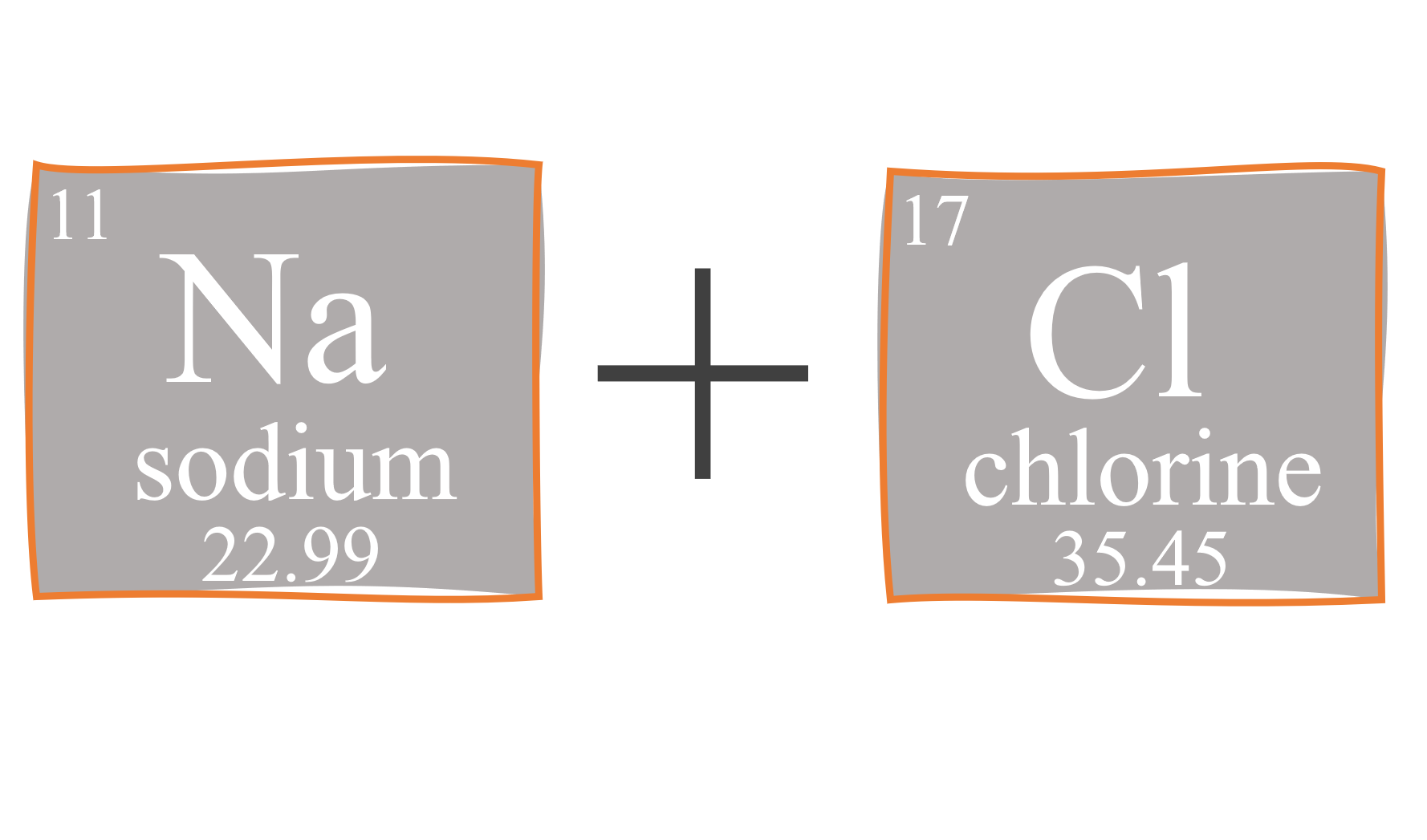
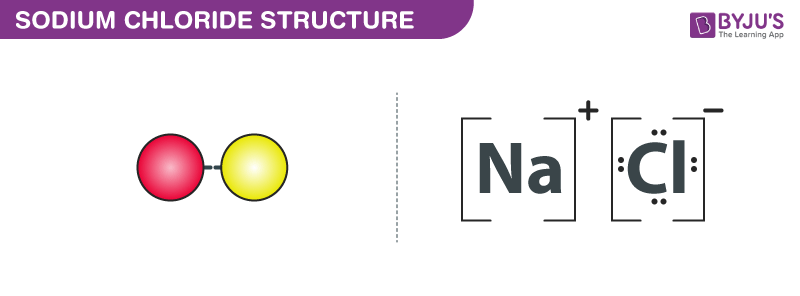
:max_bytes(150000):strip_icc()/sodium-chloride-structure-artwork-160936423-589330f15f9b5874eea7ba04.jpg)