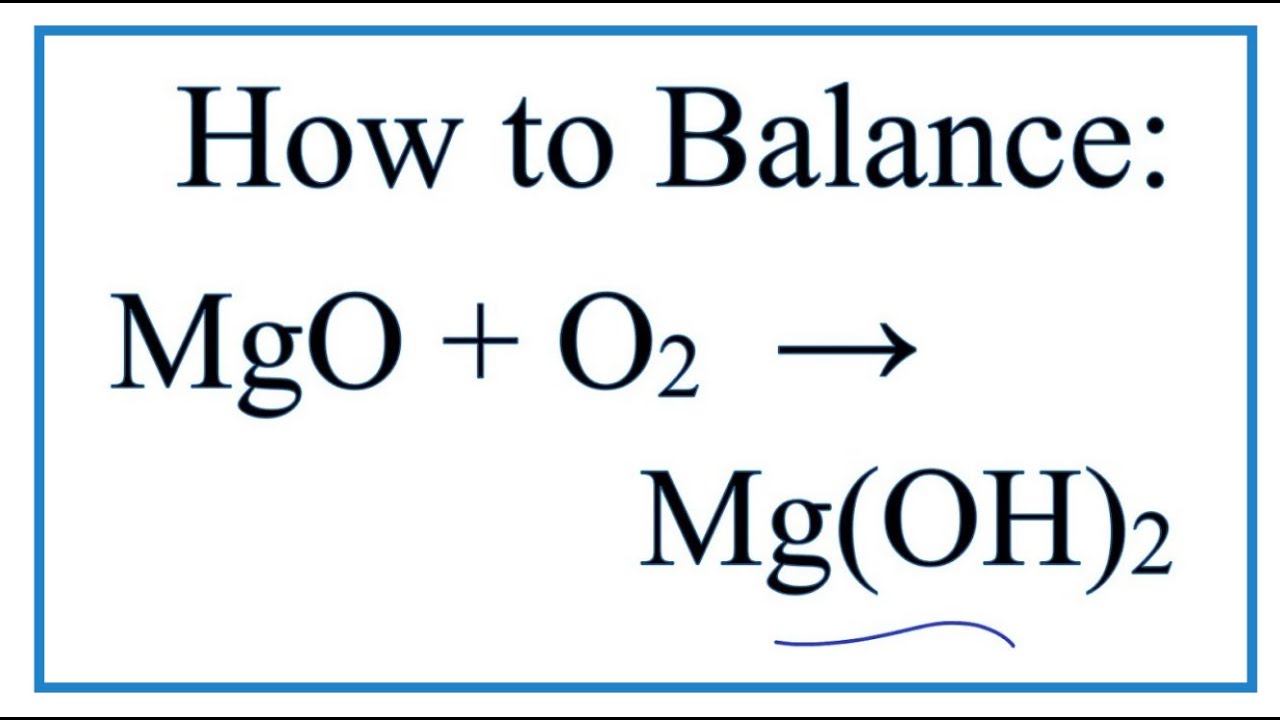A 3.250g sample of magnesium is burned in a container of 12.500g oxygen. What mass of oxygen gas remains unreacted after the magnesium has been completely consumed to form magnesium oxide as

Ionic equations A chemical equation shows the number of atoms and molecules of the reactants and products. Also shows physical state of reactants and products. - ppt download

Question Video: Calculating Oxidation State Change for Magnesium during Magnesium Combustion | Nagwa

Metal carbonates are known to undergo thermal decomposition, producing the metal oxide and releasing carbon dioxide. The process is described by the following generic equation, in which M represents an unknown divalent
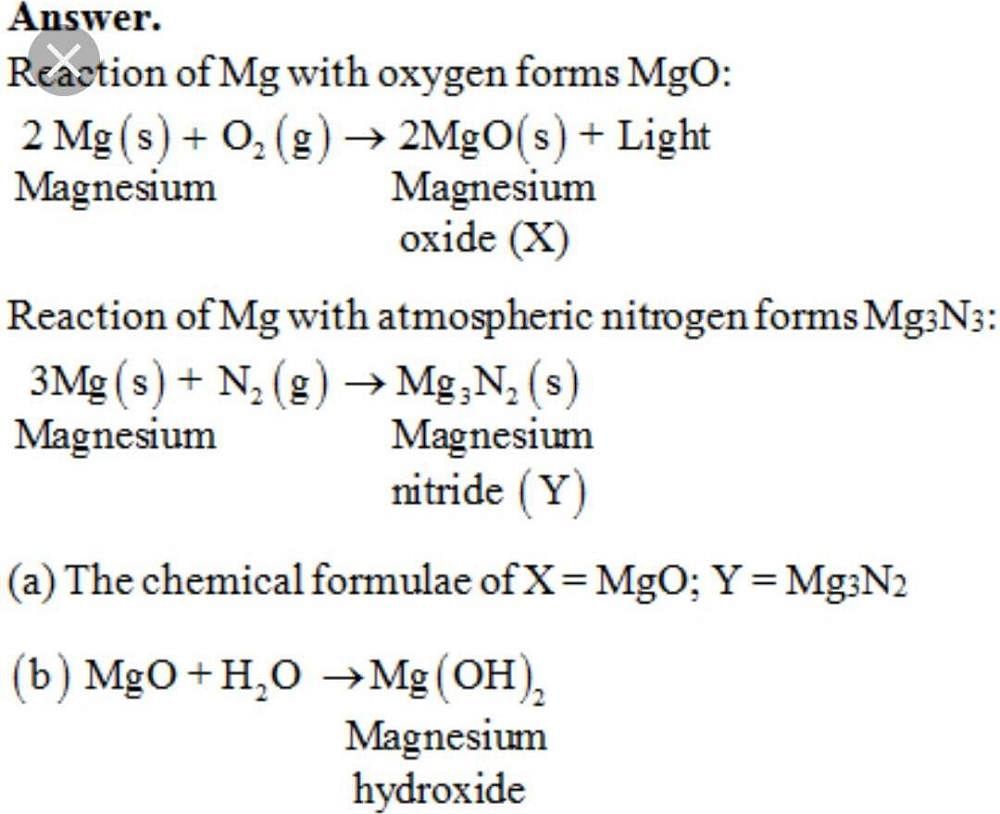
Explain the action of dil.HCL on the following with chemical equations 1- Magnesium ribbon 2-Sodium hydroxide 3-Crushed egg shells? | EduRev Class 10 Question

CHEMICAL EQUATIONS. Chemical equations You are expected to know these names and formula: NameFormulaNameFormula WaterH2OH2OHydrchloric Acid HCl Carbon. - ppt download
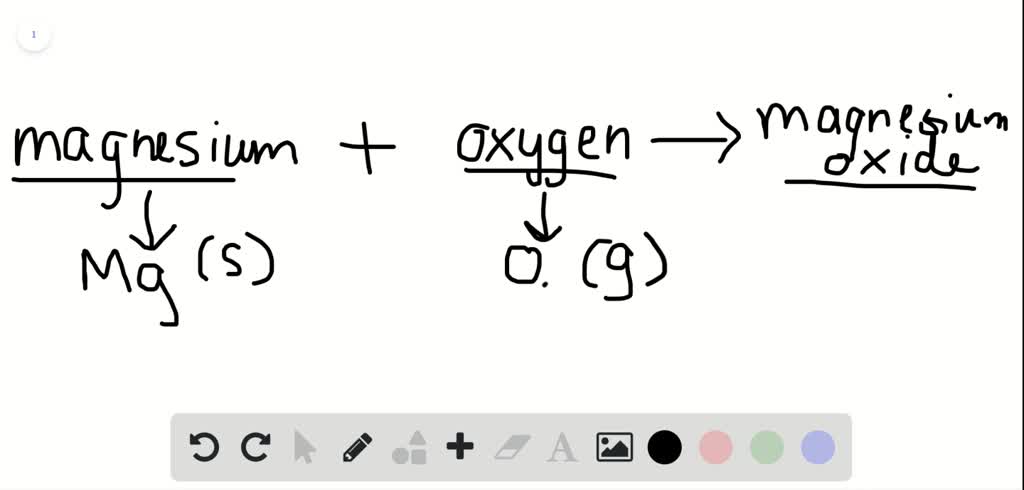
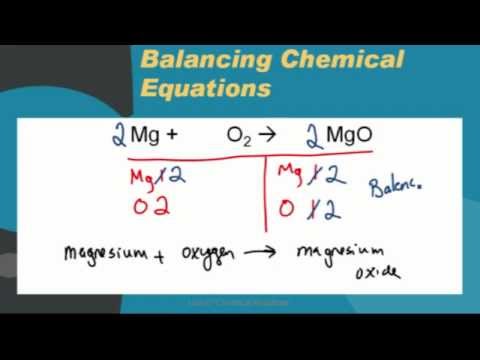
SOLVED:Magnesium oxide forms from magnesium metal and oxygen gas. Write a word equation and an unbalanced formula equation. Include all of the appropriate notations.
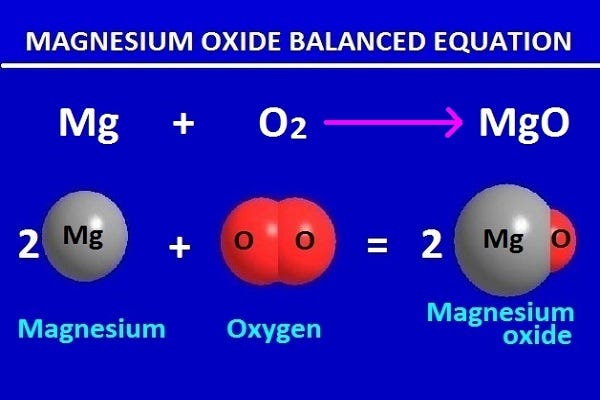
Magnesium oxide balanced equation in chemistry for class 9 | by KAKALI GHOSH , Teacher,blogger. M.Sc chemistry. | Medium

Question Video: Identifying the Oxidized Specie in the Reaction of Magnesium Oxide with Hydrogen | Nagwa

Explain the formation of magnesium oxide from magnesium and oxygen? Analyze the electron dot diagram and complete the table.

SOLVED: Use the formula of magnesium oxide (MgO) to write out a balanced chemical equation of the burning of magnesium metal in oxygen gas to generate magnesium oxide. Make sure to indicate