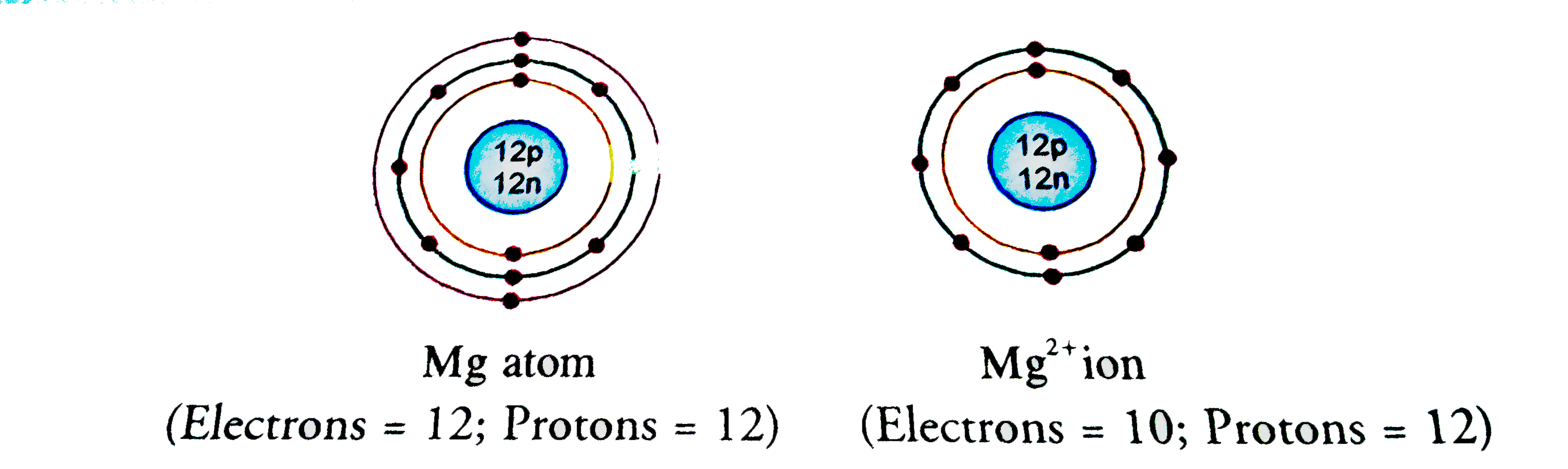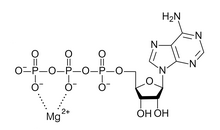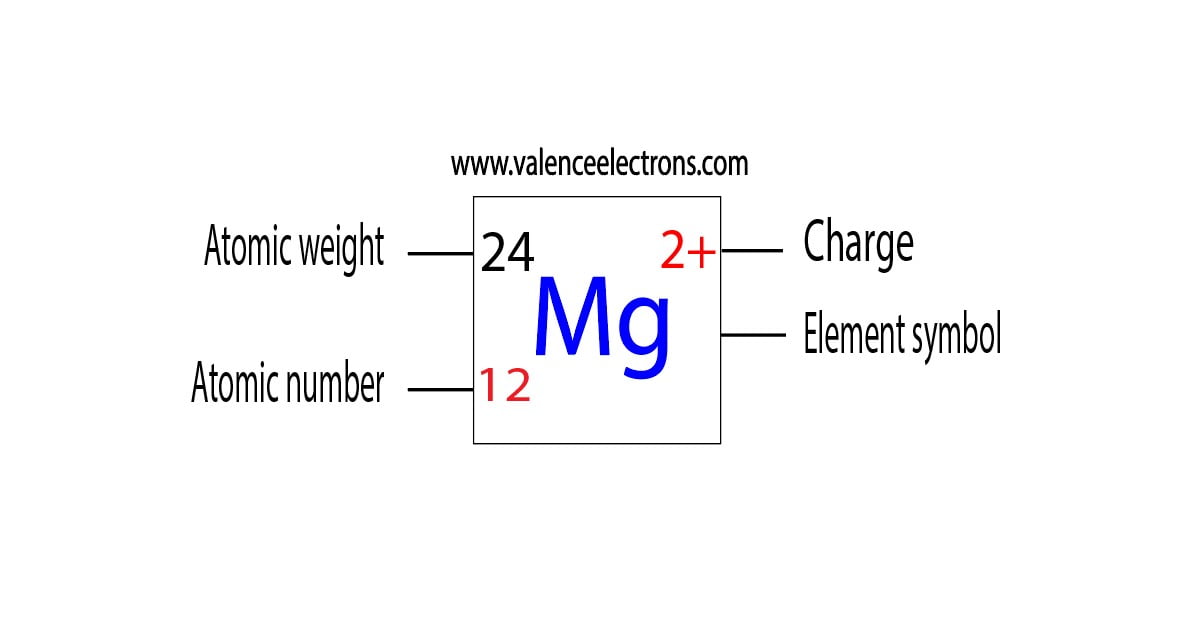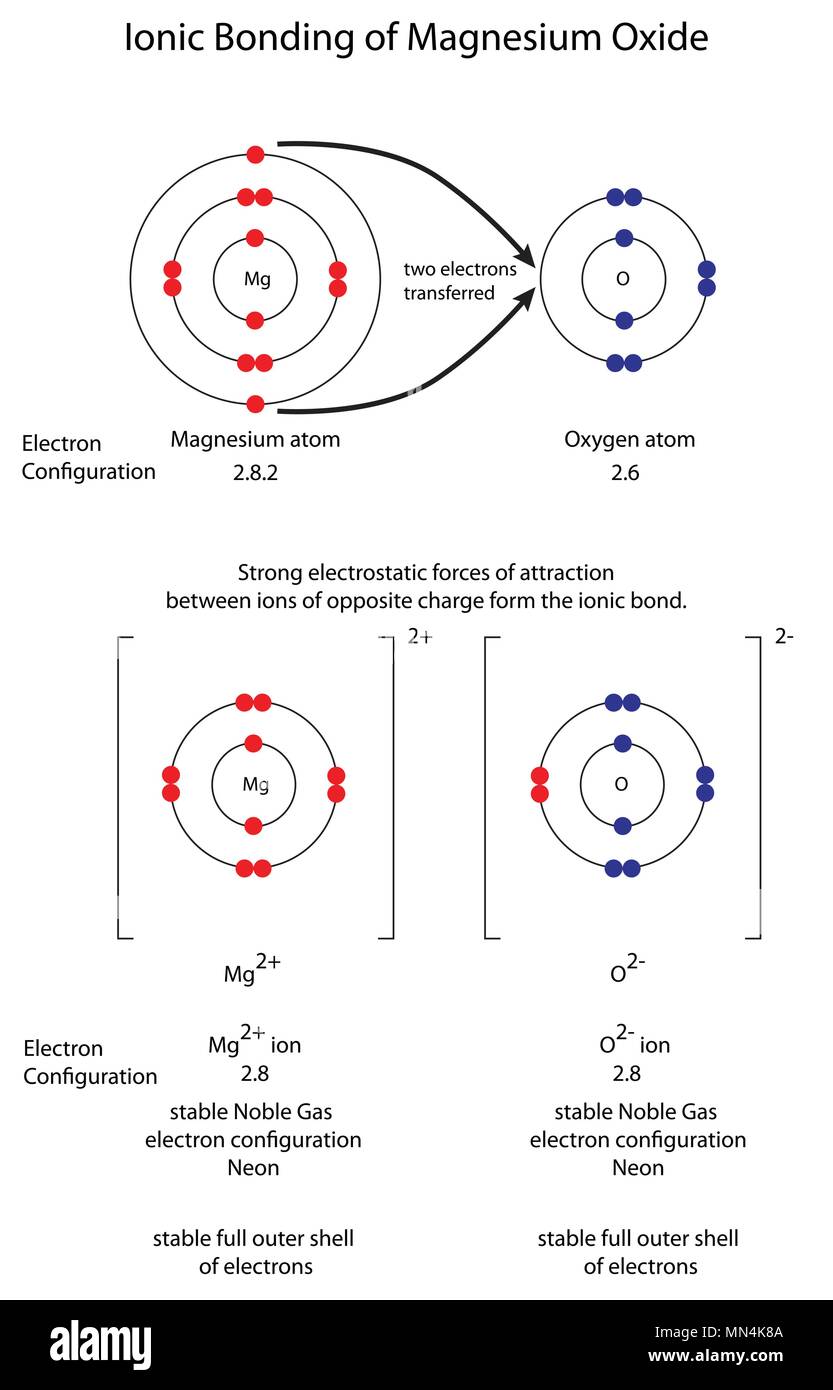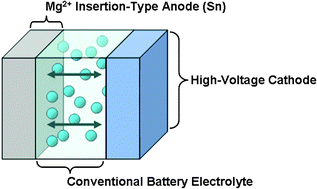
A high energy-density tin anode for rechargeable magnesium-ion batteries - Chemical Communications (RSC Publishing)

Magnesium-ion batteries for electric vehicles: Current trends and future perspectives - Raj Shah, Vikram Mittal, Eliana Matsil, Andreas Rosenkranz, 2021

High‐Rate and Long Cycle‐Life Alloy‐Type Magnesium‐Ion Battery Anode Enabled Through (De)magnesiation‐Induced Near‐Room‐Temperature Solid–Liquid Phase Transformation - Wang - 2019 - Advanced Energy Materials - Wiley Online Library

SOLVED: Question 5 (3 points) What is the formula for the ionic compound formed by magnesium ion and hypochlorite ion? OMg(ClO)2 OMgClO OMgClO2 OMg(ClO2)2

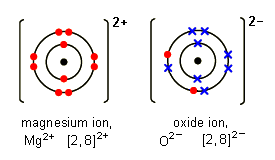
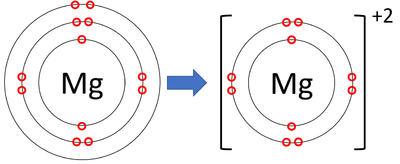
Question Video: Identifying the Energy Level Diagram That Represents the Electronic Configuration of the Magnesium Ion | Nagwa

Importance of Metal Hydration on the Selectivity of Mg2+ versus Ca2+ in Magnesium Ion Channels | Journal of the American Chemical Society

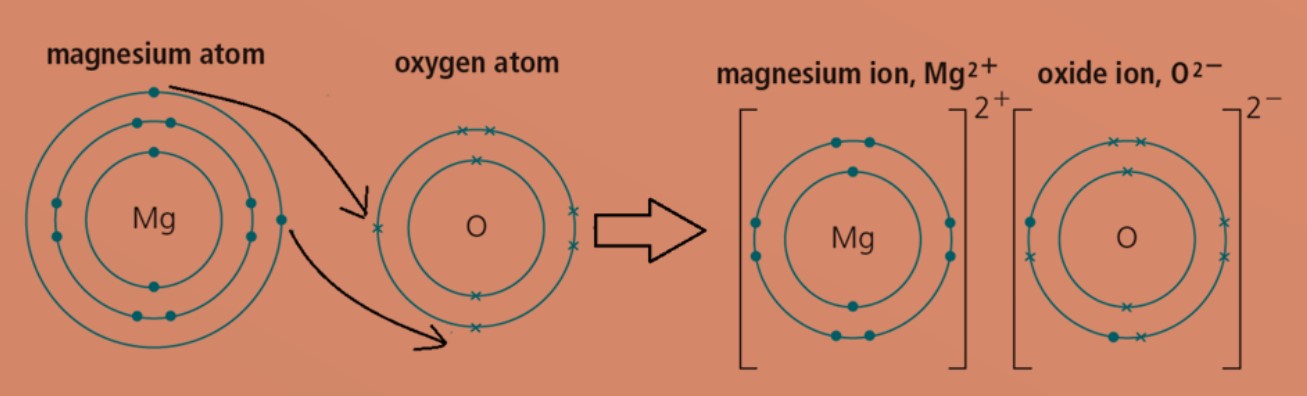
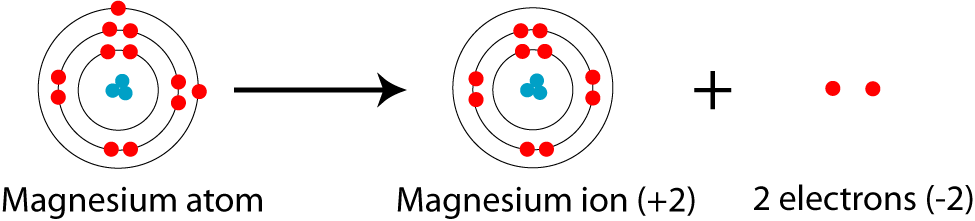
What is the Difference Between Magnesium Atom and Magnesium Ion | Compare the Difference Between Similar Terms
Hat is the charge of the ion most likely to be formed from Magn to achieve noble gas notation? | Socratic

Based on the octet rule, magnesium most likely forms a ion. a. Mg2- b. Mg2+ c. Mg- d. Mg6- e. Mg6+ | Homework.Study.com

Effect of magnesium ion on sylvite flotation: An experiment and molecular dynamic simulation study - ScienceDirect

What is the formula of the compound formed between the magnesium ion and the acetate ion? A. MgC2H3O2 B. Mg2C2H3O2 C. Mg(C2H3O2)2 D. Mg(C2H3O2)3 | Homework.Study.com