SOLVED: The active ingredients in a tablet of a popular antacid are listed as 585 mg of CaCO3 and 112 mg of Mg(OH): These each react with hydrochloric acid found in your

For Your Research. The Four Research Questions 1.What is the chemistry (including an equation) of the process? 2.What are the factors that impact on the. - ppt download

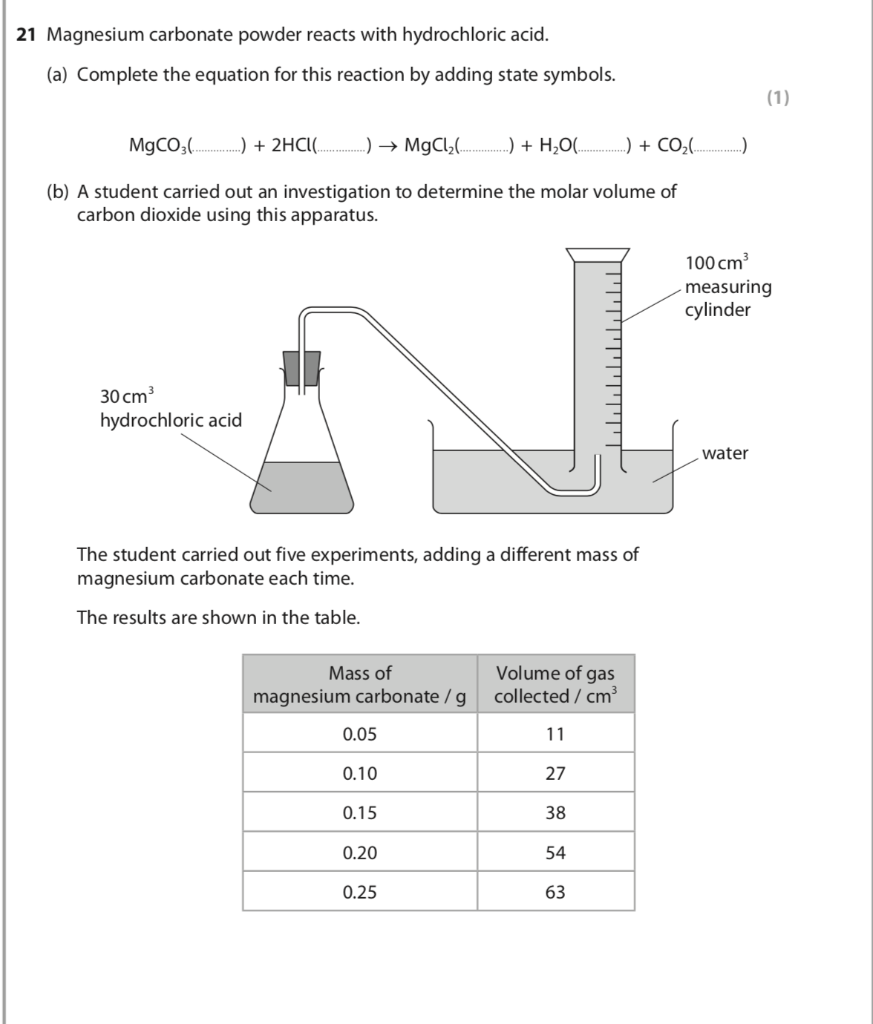
5 g of a sample of magnesium carbonate on treatment with excess of dilute hydrochloric acid gave 1.12 L of CO(2) at STP . The percentage of magnesium carbonate in the mixture is

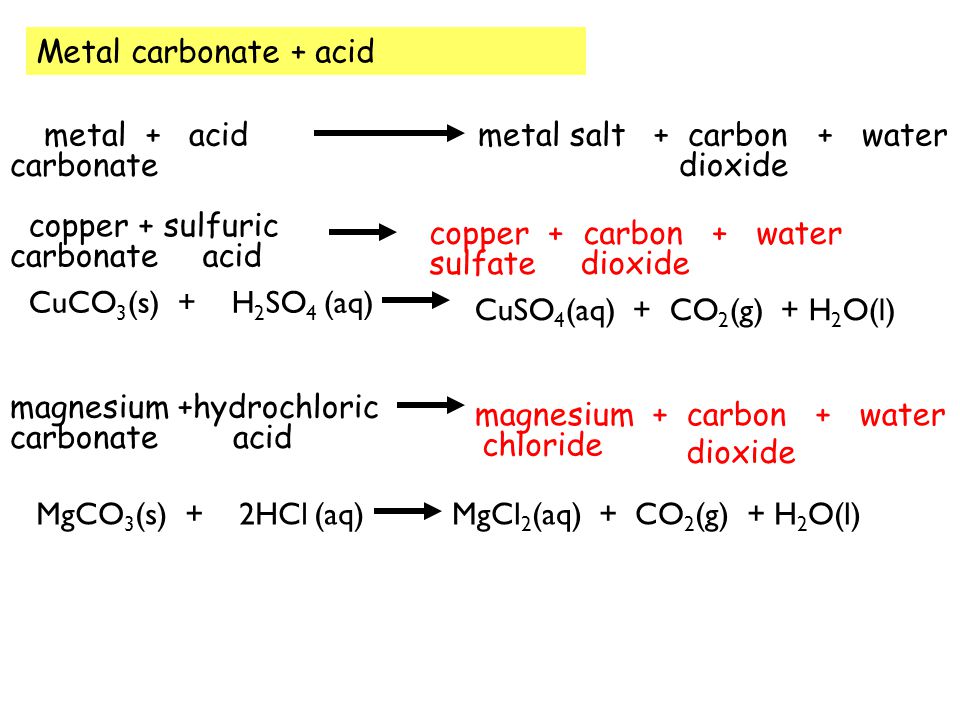
SOLVED: 6(20 Rts) ^ mixture containing Magnesium Carbonate (MgcOxs) is reacted with an excess of hydrochloric acid according to the following equation: MgCO3) 2HCltaa) MgClzea) + CO(g) HzOqu) Calculate the " percentage

filosoffen.dk - what is metformin 500 mg used for | Commit error. what is the word equation for calcium carbonate and hydrochloric acid congratulate
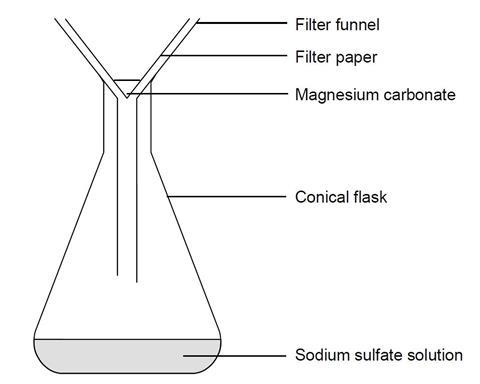
Making magnesium carbonate: the formation of an insoluble salt in water | Experiment | RSC Education