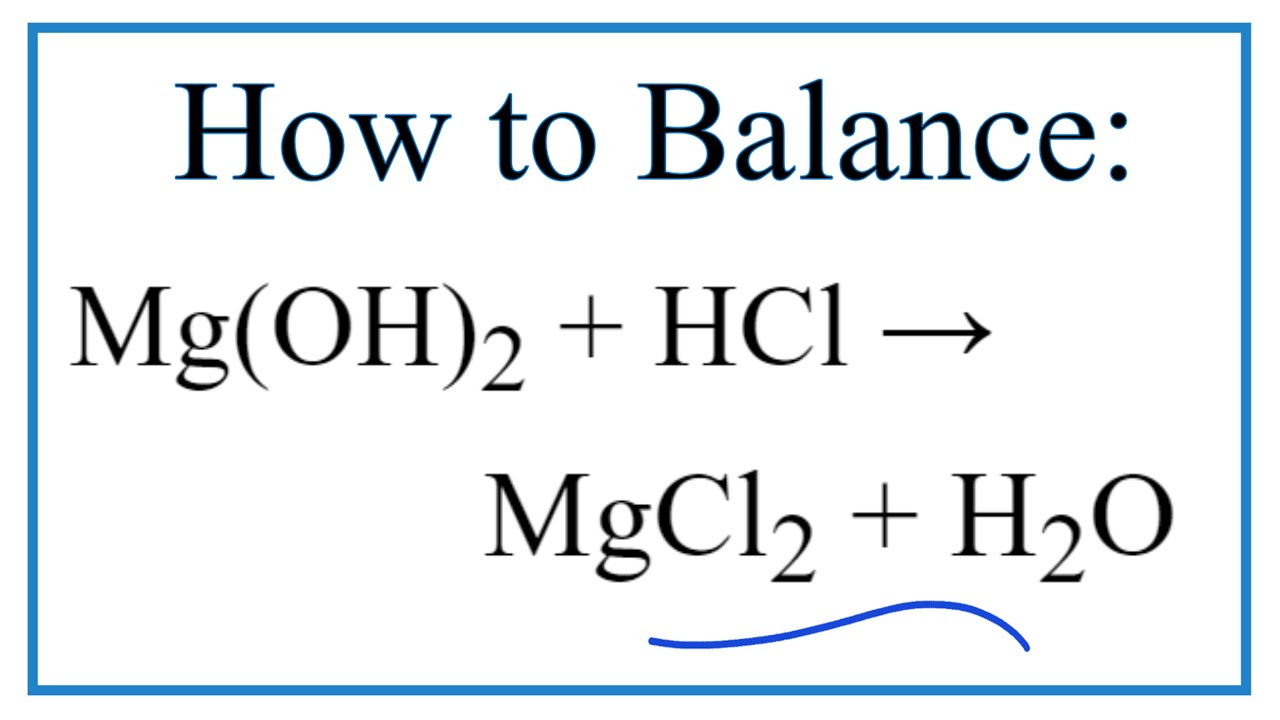
SOLVED: When hydrochloric acid reacts with magnesium hydroxide, the products that form are magnesium chloride and water. What is the coefficient for water when this equation is properly balanced?

What happens magnesium react with hydrochloric acid? - Find 3 Answers & Solutions | LearnPick Resources

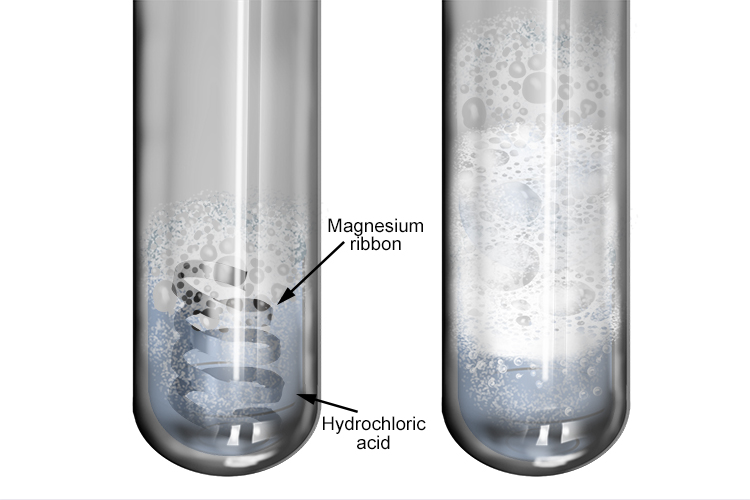
Question Video: Recognizing Why Warm Hydrochloric Acid Has a Faster Rate of Reaction with Magnesium than Cold Hydrochloric Acid | Nagwa
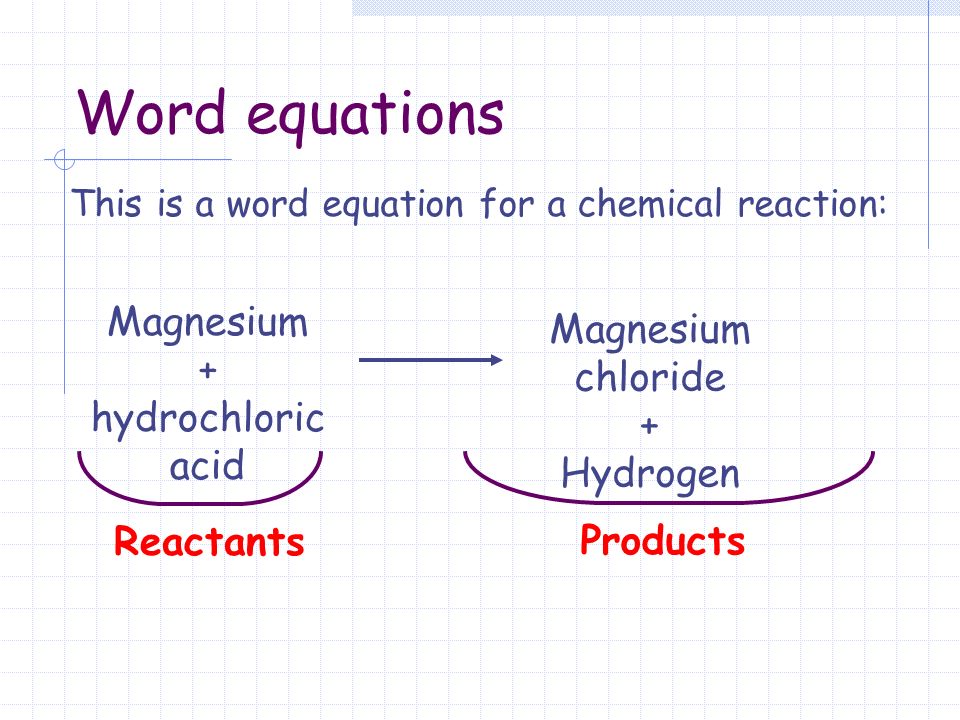
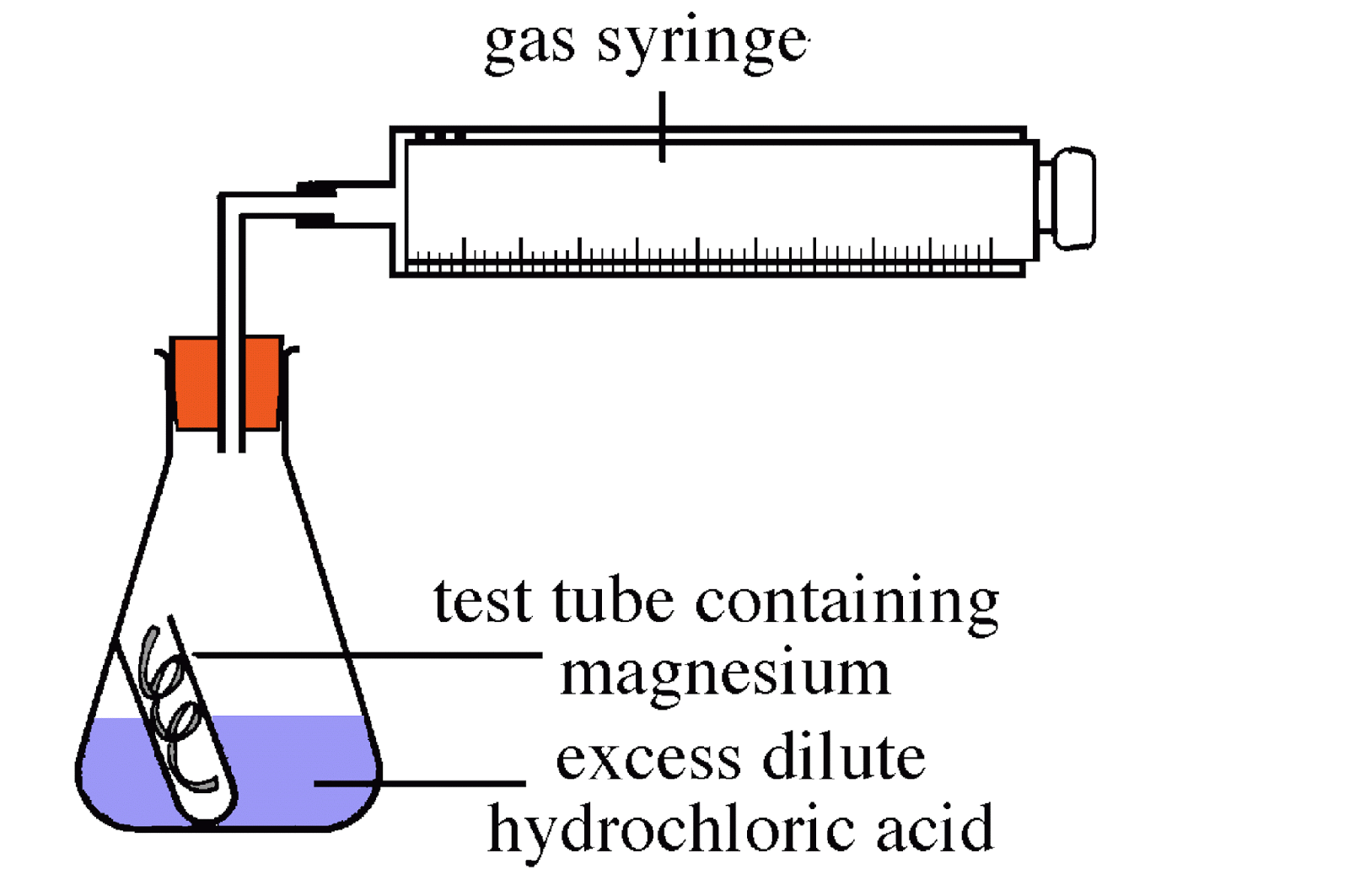
This is a word equation for a chemical reaction: Reactants Products Magnesium + hydrochloric acid Magnesium chloride + Hydrogen Word equations. - ppt download

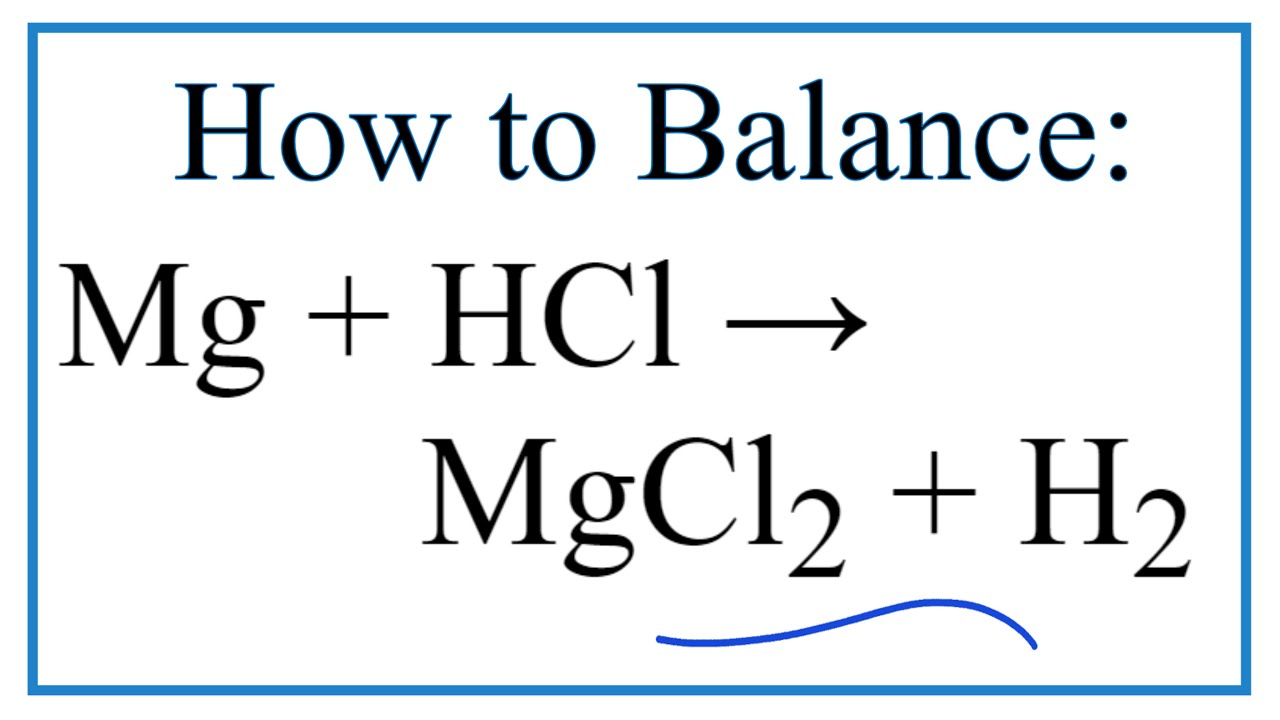
How to Balance Mg + HCl → MgCl2 + H2 (Magnesium + Hydrochloric Acid) | How to Balance Mg + HCl → MgCl2 + H2 (Magnesium + Hydrochloric Acid) One of the