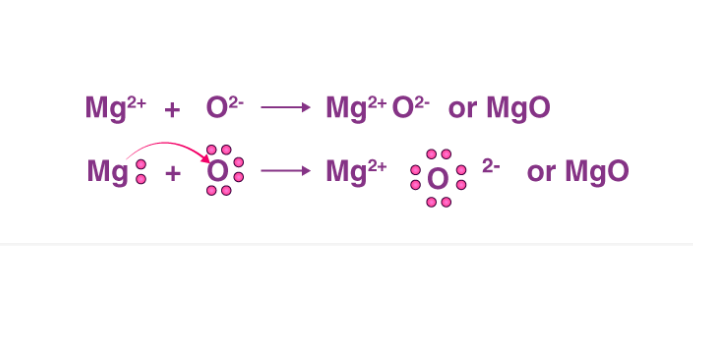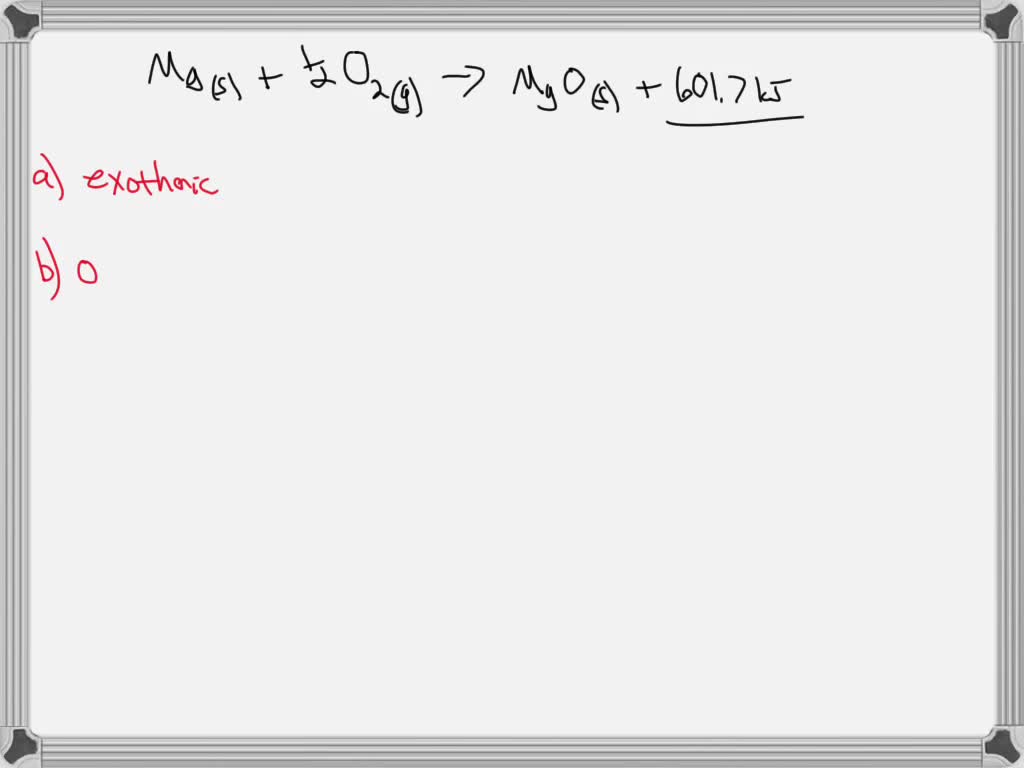
SOLVED: The formation of magnesium oxide, MgO, is shown below: Mg(s) + 1/2 O2( g) → MgO(s) + 601.7 kJ a) Is this an endothermic or exothermic reaction? b) If 0.321 moles
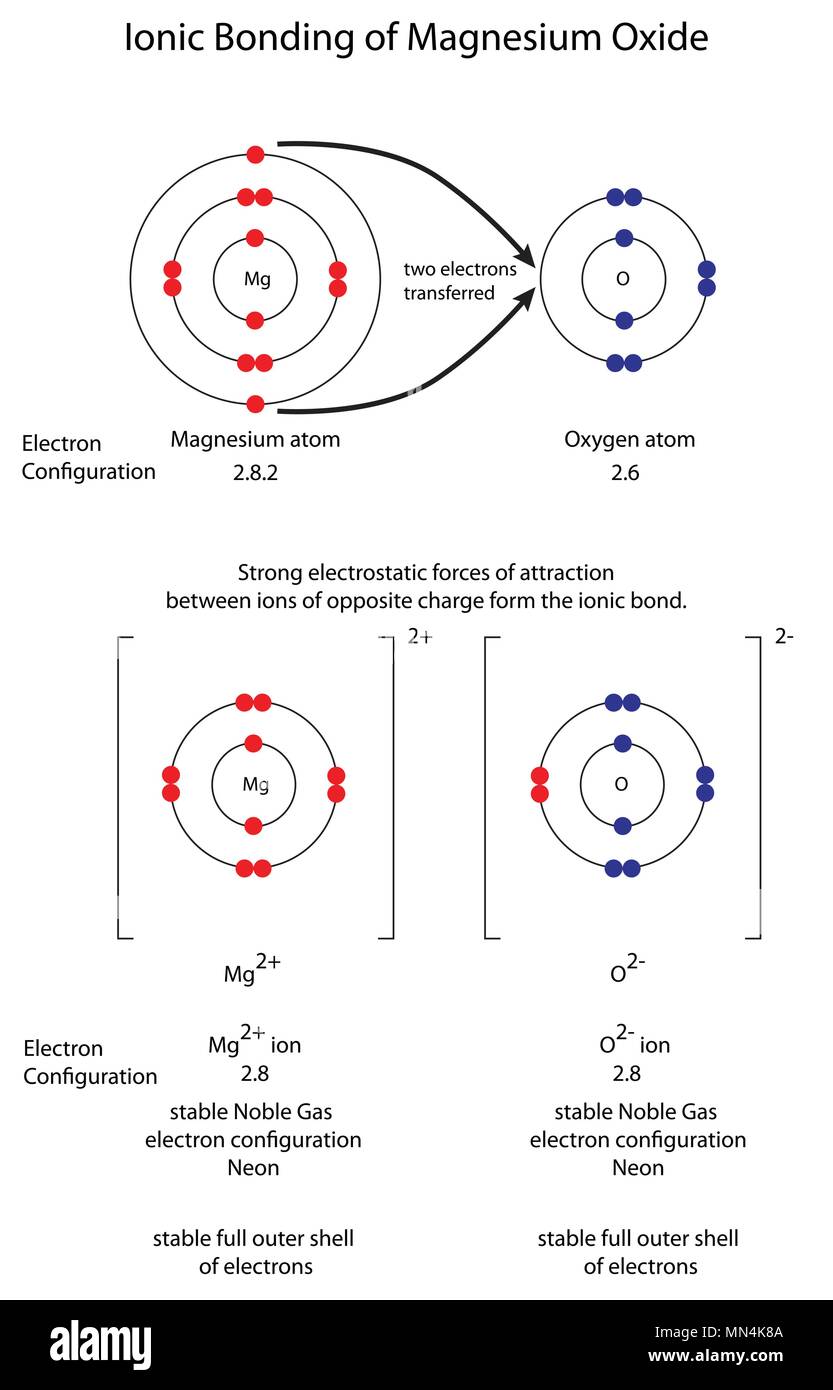
![Answered] Show the formation of Mgo by the transfer of electrons in the two elements using electron - Brainly.in Answered] Show the formation of Mgo by the transfer of electrons in the two elements using electron - Brainly.in](https://hi-static.z-dn.net/files/d1a/3a413915efd0da13a1cf9e6327e61aaa.jpg)
Answered] Show the formation of Mgo by the transfer of electrons in the two elements using electron - Brainly.in

Answer the following a)Write the electron - dot structures for sodium, oxygen and magnesium b) Show the formation of - Science - - 13004889 | Meritnation.com
Experiment 7: Enthalpy of Formation of Magnesium Oxide Purpose: In this experiment, Hess' law will be used to determine the ent