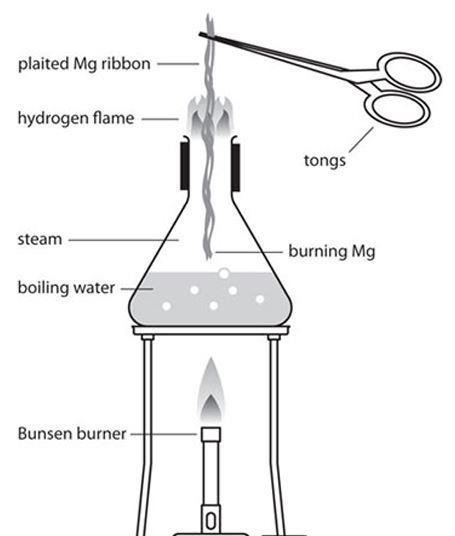
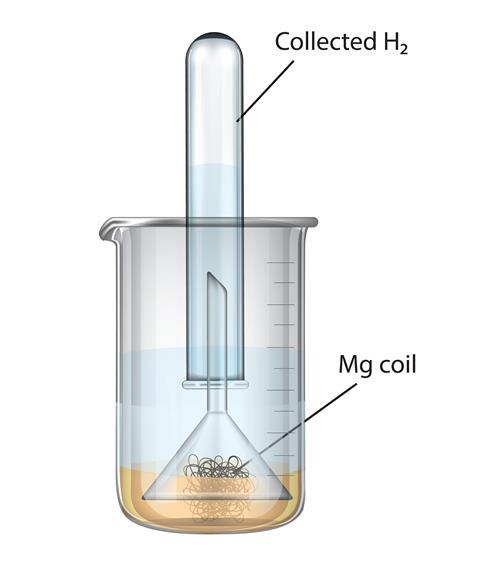
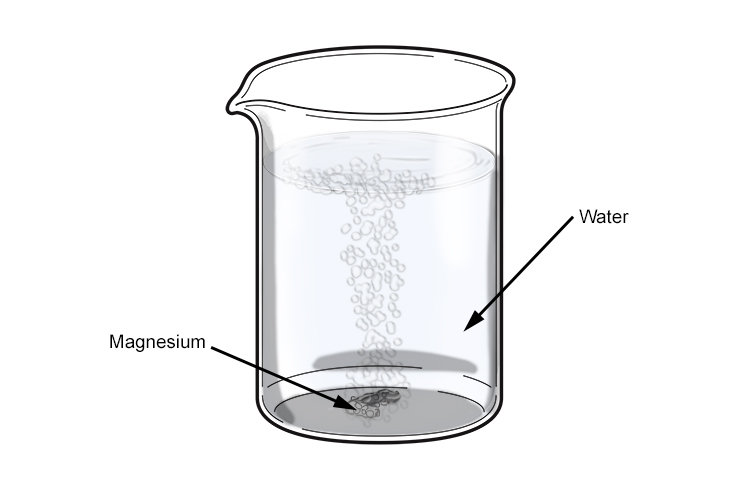
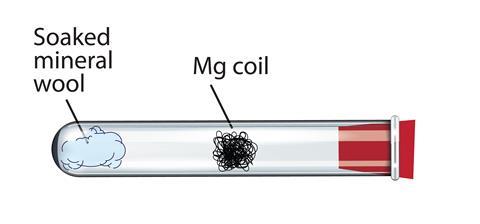
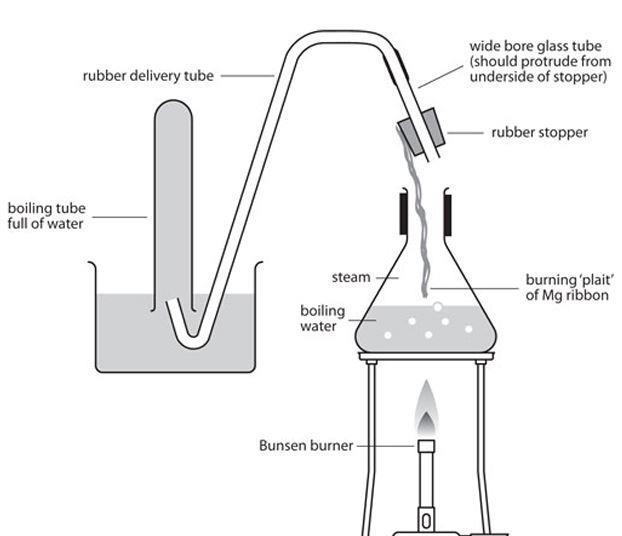
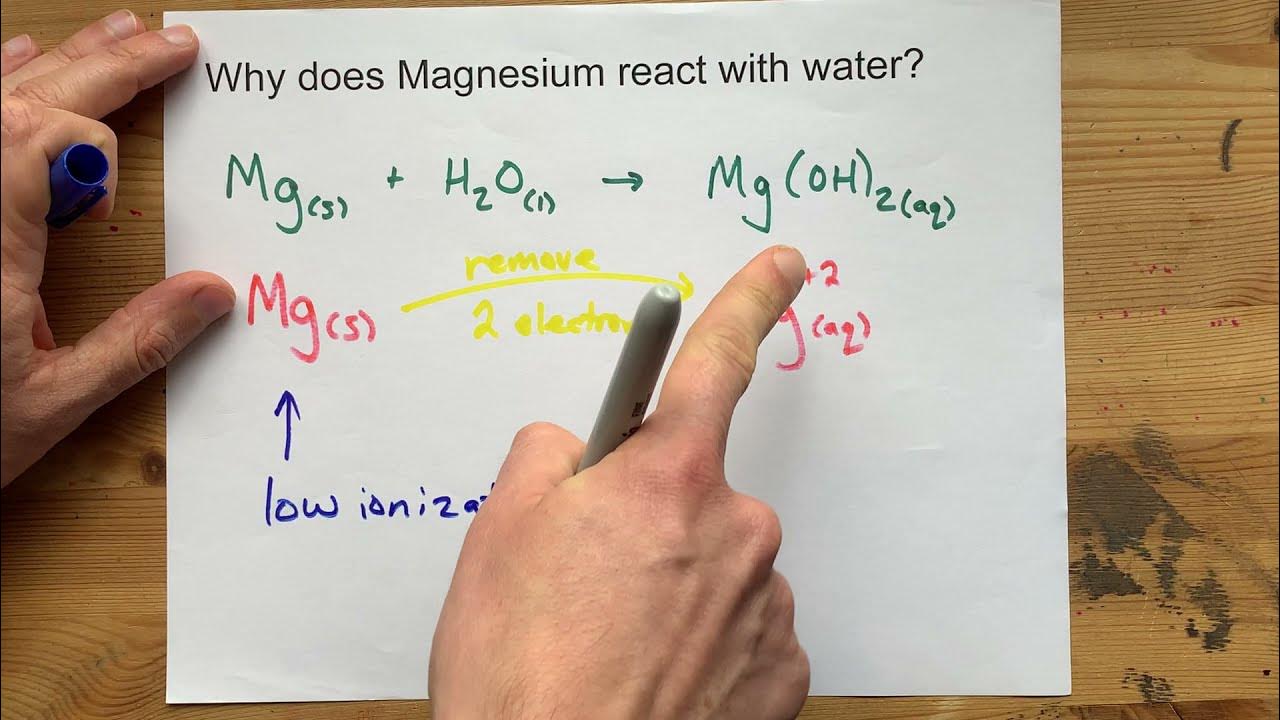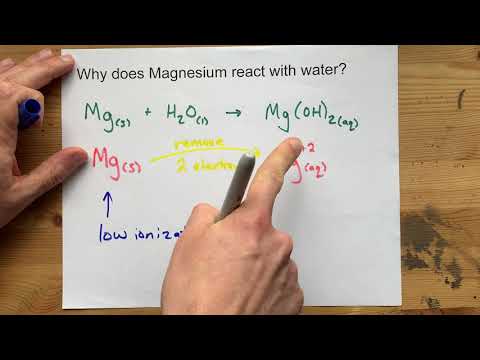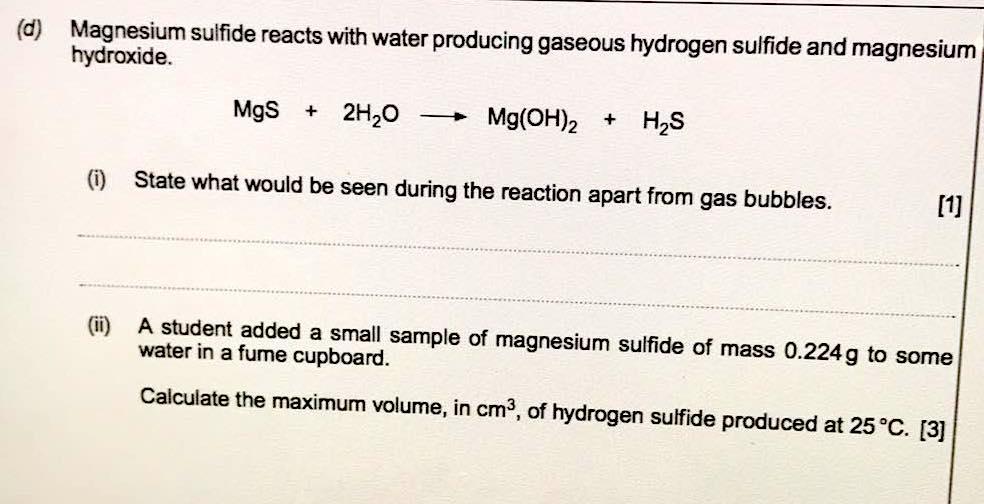why calcium and magnesium floats on water - Science - Metals and Non-metals - 13061069 | Meritnation.com

Metals react differently with water. The more vigorous is the reaction, the more is the bubble formation. The following diagram illustrates the reaction of sodium, magnesium and copper with water.What can be

Question Video: Ordering the Reactions of Magnesium Metal with Varying Concentrations of Nitric Acid | Nagwa

SOLVED: Magnesium reacts with water in a single replacement reaction. Which reaction shows the correctly balanced equation? A. Mg + H2O -> MgO + H2 B. Mg + H2O -> 2MgO +