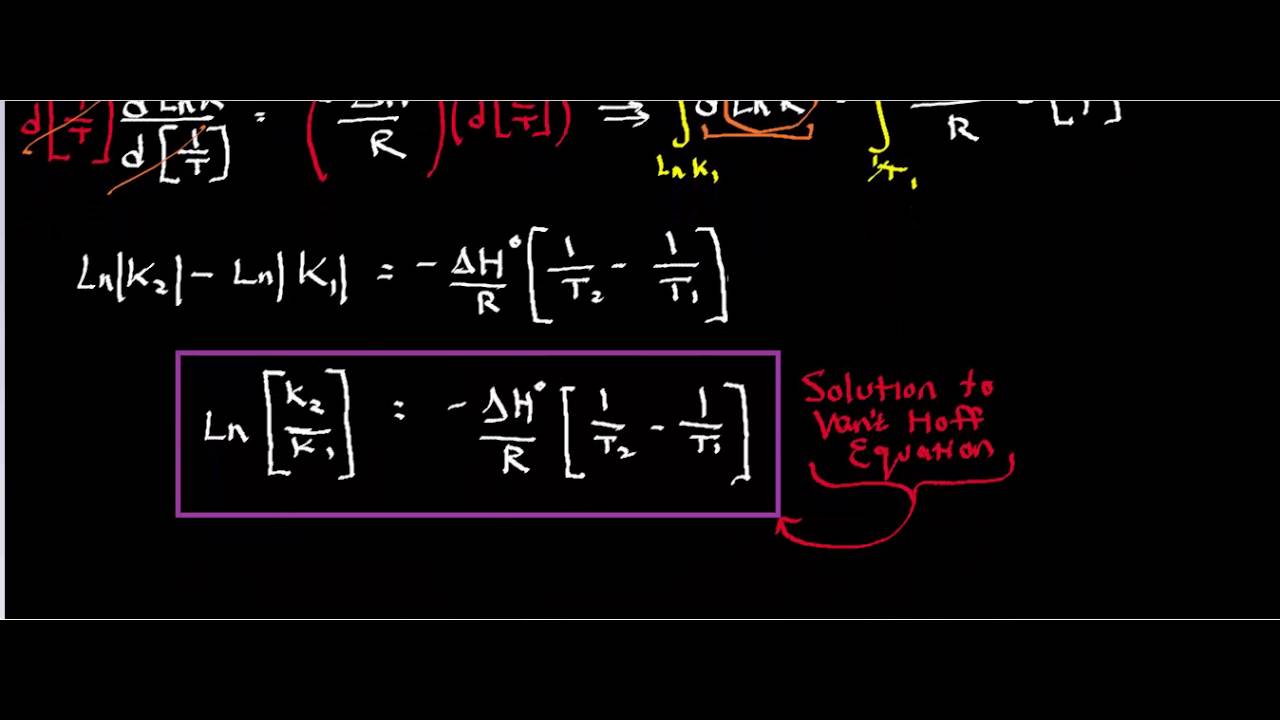
SOLVED: Membrane Separation II Reverse Osmosis (a) Derive van Hoff' s equation (osmotic pressure T = cRT) (b) feed solution containing Sg NaCI/L (p-1OOlkg/m ) is purified using reverse osmosis system: Water
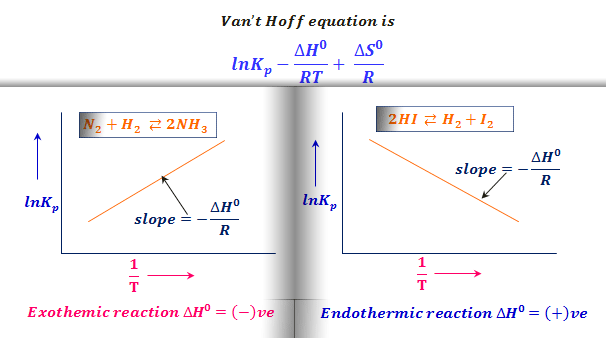
Van't Hoff equation-equilibrium. How much Van't Hoff equation — effect… | by Chemistry Topics | Medium

PDF) Van't Hoff equation-equilibrium How much Van't Hoff equation -effect on temperature | Kajal Panda - Academia.edu

SOLVED: a) The van't Hoff equation: A,Ho 4,So In K = RT relates the change in temperature to the chemical equilibrium constant Derive the van't Hoff equation starting with the expression for