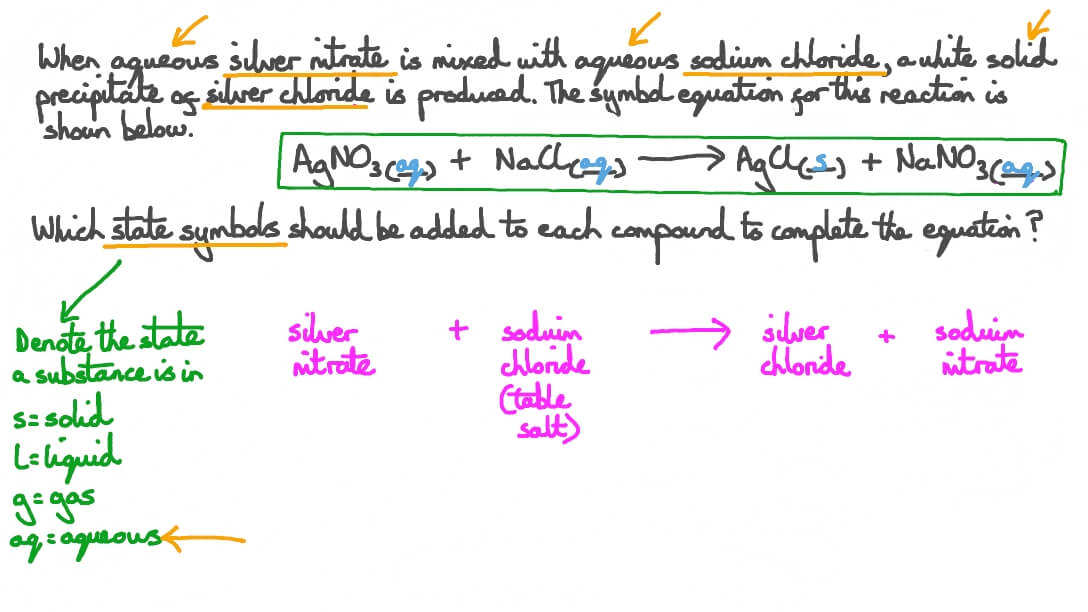
Question Video: Symbol Equation for the Reaction of the Precipitation of Agno3 with State Symbols | Nagwa

Factory Supply Grade Chemicals Ar Gr Grade Agno3 Colorless and Transparent Crystal 99.8% Silver-Nitrate - China Silver-Nitrate Electroplating Grade, Silver-Nitrate Solid | Made-in-China.com
✓ Solved: Valence, Bonding, and Lewis Structures When a solution of salt (sodium chloride) in water is...
A solution of common salt when added to silver nitrate solution yields a precipitate of silver chloride (0.28 g). - Sarthaks eConnect | Largest Online Education Community

SOLVED: Text: Preparation of Rochelle Salt Suppose you are hired to haul away the waste left over from a place that develops film. It turns out the waste actually contains a lot

A galvanic cell is made up of a copper electrode in a 1.0 M copper (II) sulfate solution, a silver electrode in a 1.0 M silver nitrate solution, and a salt bridge