SOLVED: The rate constant for the first-order isomerization of cyclopropane C3H6 to propane CH3CH=CH2 is 6.7 x 10^-4 s^-1 at 500 degrees celsius. What is its value at 300 degrees celsius? Ea=272

SOLVED: calculate the value of heat of vaporization of ice at-1c.The values of the pressure of water vapour are 3.88mm hg and 4.58 mm hg at 0c
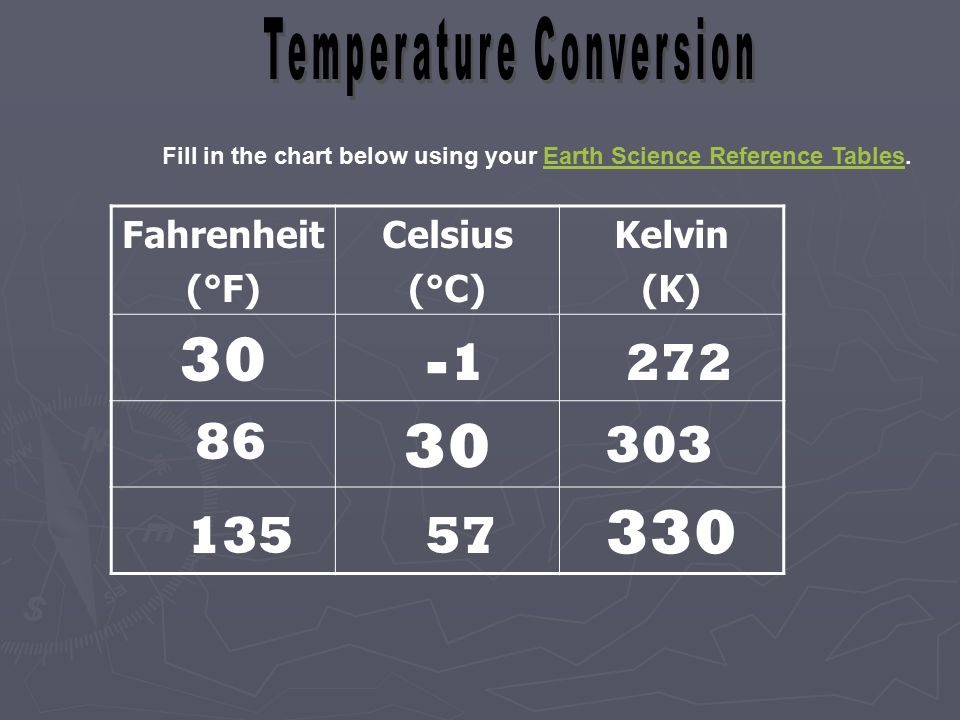
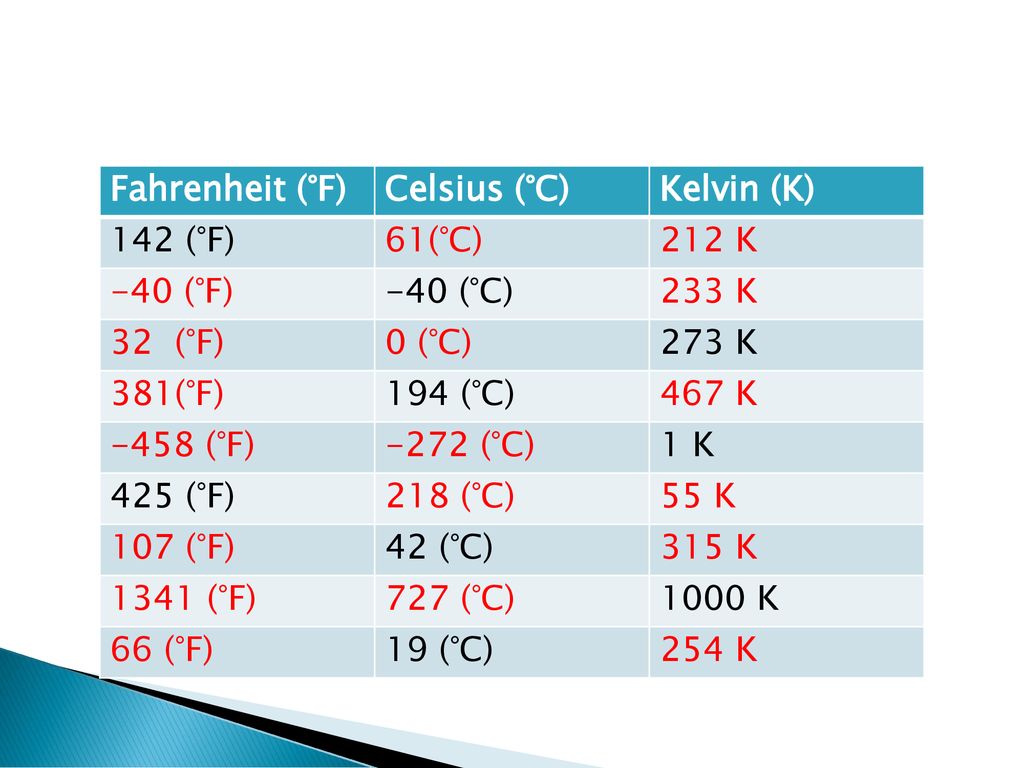
Joules and Water. Fahrenheit (°F) Celsius (°C) Kelvin (K) Fill in the chart below using your Earth Science Reference Tables.Earth Science Reference. - ppt download

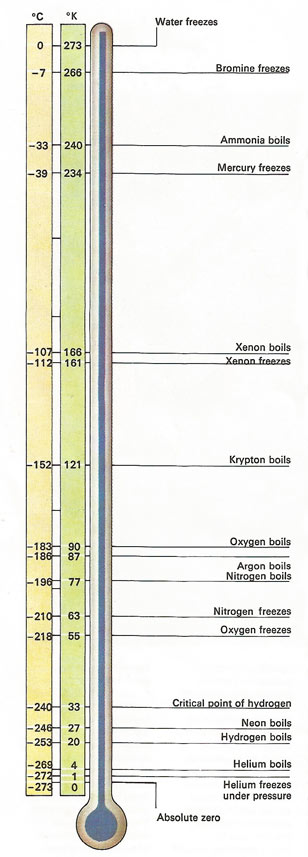
Kelvin Scale vs Celsius scale nothing can get colder than -273 degrees Celsius - LIACOS EDUCATIONAL MEDIA

If H C , H K and H F are heat required to raise the temperature of one gram of water by one degree in Celsius,Kelvin and Fahrenheit temperature scales respectively then : -
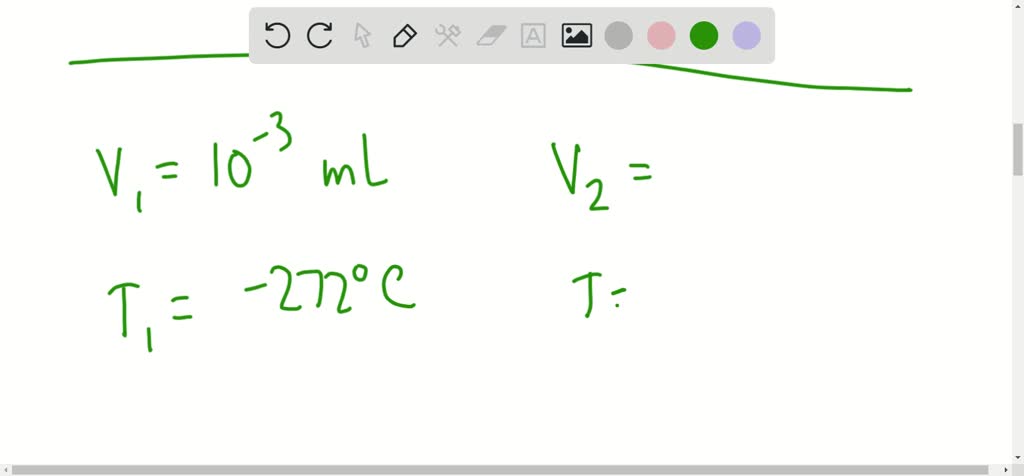
SOLVED:For each of the following sets of volume/temperature data, calculate the missing quantity. Assume that the pressure and the amount of gas remain constant. a. V=22.4 L at 0^∘ C ; V=44.4

Conversion of Kelvin to Degree Celsius | How to convert Kelvin to degree Celsius | Kelvin in Celsius - YouTube